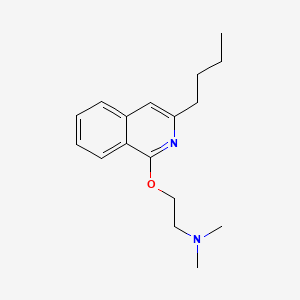
Drug
D0105 | Quinisocaine
D
D04AB05 Quinisocaine
[D04AB] Anesthetics for topical use
[D04A] ANTIPRURITICS, INCL. ANTIHISTAMINES, ANESTHETICS, ETC.
[D04] ANTIPRURITICS, INCL. ANTIHISTAMINES, ANESTHETICS, ETC.
[D] Dermatological drugs
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| OXYGEN CONSUMPTION RATE (OCR) | 300 μM | 2 minutes | human | HepG2 | Measurement of OCR | Negative | EC50 | 7 |
| ECAR | 300 μM | 2 minutes | human | HepG2 | Measurement of ECAR | Negative | EC50 | 7 |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| quail | LD50 | oral | > 100mg/kg (100mg/kg) | Ecotoxicology and Environmental Safety. Vol. 6, Pg. 149, 1982. | |
| rat | LD50 | unreported | 2300mg/kg (2300mg/kg) | Drugs in Japan Vol. 6, Pg. 721, 1982. | |
| mouse | LD50 | subcutaneous | 3500mg/kg (3500mg/kg) | Takeda Kenkyusho Ho. Journal of the Takeda Research Laboratories. Vol. 30, Pg. 530, 1971. | |
| mouse | LD50 | intravenous | 2500mg/kg (2500mg/kg) | Takeda Kenkyusho Ho. Journal of the Takeda Research Laboratories. Vol. 30, Pg. 530, 1971. | |
| rat | LD50 | intraperitoneal | 1500mg/kg (1500mg/kg) | Advances in Teratology. Vol. 3, Pg. 181, 1968. | |
| rat | LD50 | oral | 8400mg/kg (8400mg/kg) | Iyakuhin Kenkyu. Study of Medical Supplies. Vol. 4, Pg. 467, 1973. | |
| rat | LD50 | intravenous | 2320mg/kg (2320mg/kg) | vascular: other changes | Takeda Kenkyusho Ho. Journal of the Takeda Research Laboratories. Vol. 30, Pg. 735, 1971. |
| mouse | LD50 | intraperitoneal | 3050mg/kg (3050mg/kg) | Takeda Kenkyusho Ho. Journal of the Takeda Research Laboratories. Vol. 30, Pg. 530, 1971. | |
| rat | LD50 | subcutaneous | 3900mg/kg (3900mg/kg) | Takeda Kenkyusho Ho. Journal of the Takeda Research Laboratories. Vol. 30, Pg. 530, 1971. | |
| mouse | LD50 | oral | 9100mg/kg (9100mg/kg) | Takeda Kenkyusho Ho. Journal of the Takeda Research Laboratories. Vol. 30, Pg. 530, 1971. | |
| 1-(.beta.-Dimethylaminoethoxy)-3-N-butylisoquinoline | 1-(2-Dimethylaminoethoxy)-3-butylisochinolin | 2-(Dimethylamino)ethoxy-3-butyl-isoquinoline |
| 2-[(3-Butyl-1-isoquinolinyl)oxy]-N,N-dimethylethanamine | 2-[(3-Butyl-1-isoquinolinyl)oxy]-N,N-dimethylethanamine # | 2-[(3-Butylisoquinolin-1-yl)oxy]-N,N-dimethylethan-1-amine |
| 3-Butyl-1-(2-dimethylaminoethoxy)isoquinoline | 3-Butyl-1-[2-(dimethylamino)ethoxy]isoquinoline | 3-Butyl-1-[2-(dimethylamino)ethyl]isoquinoline |
| 5-21-03-00414 (Beilstein Handbook Reference) | 772EN3BH6I | 86-80-6 |
| AB00514685 | AKOS022143560 | AO-080/43342670 |
| BDBM50225699 | BPBio1_000660 | BRD-K73391359-003-03-7 |
| BRN 0018179 | BSPBio_000600 | CAS-2773-92-4 |
| CHEBI:93295 | CHEMBL127643 | Chinisocain |
| Chinisocainum | D08462 | DTXSID8048525 |
| Dimethisoquin | EINECS 201-700-0 | Ethanamine, 2-((3-butyl-1-isoquinolinyl)oxy)-N,N-dimethyl- |
| Ethanamine, 2-[(3-butyl-1-isoquinolinyl)oxy]-N,N-dimethyl- | Histaderme | ISOQUINOLINE, 3-BUTYL-1-(2-(DIMETHYLAMINO)ETHOXY)- |
| Isochinol | Isoquinoline, 3-butyl-1-[2-(dimethylamino)ethoxy]- | Isoquinoline, 3-butyl-1-[2-(dimethylamino)ethyl]- |
| Kinisokain | LS-85370 | MCULE-8714132699 |
| N-{2-[(3-butyl-1-isoquinolinyl)oxy]ethyl}-N,N-dimethylamine | NCGC00016618-01 | NCGC00016618-02 |
| NSC-39695 | NSC39695 | Prestwick0_000630 |
| Prestwick1_000630 | Prestwick2_000630 | Prestwick3_000630 |
| Prulantex | Pruralgan | Pruralgin |
| Q1478199 | Quinisocain | Quinisocaina |
| Quinisocaina [INN-Spanish] | Quinisocaine | Quinisocaine (INN) |
| Quinisocaine [INN:BAN] | Quinisocainum | Quinisocainum [INN-Latin] |
| Quinoleine | SCHEMBL8191 | SPBio_002819 |
| ST076784 | UNII-772EN3BH6I | WLN: T66 CNJ BO2N1&1 D4 |
| XNMYNYSCEJBRPZ-UHFFFAOYSA-N | ZINC1671507 | [2-(3-butylisoquinolyloxy)ethyl]dimethylamine |
| quinolein |
| DrugBank Name | Quinisocaine |
| DrugBank | DB13683 |
| CAS Number | 2773-92-4, 59-14-3, 86-80-6 |
| PubChem Compound | 6857 |
| KEGG Drug | D08462 |
| ChEBI | 93295 |
| ChemSpider | 6596 |
| Wikipedia | Quinisocaine |
1. Chan et al. (2005)