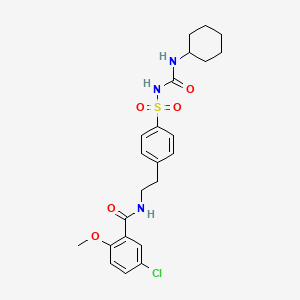
D0265 | Glibenclamide
A
A10BB01 Glibenclamide
[A10BB] Sulfonylureas
[A10B] BLOOD GLUCOSE LOWERING DRUGS, EXCL. INSULINS
[A10] DRUGS USED IN DIABETES
[A] Alimentary tract and metabolism
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| TRANSPORT OF POTASSIUM | affect | 188 | ||||||
| TRANSPORT OF POTASSIUM | K1/2 values 1-6 microM | heart and liver mitochondria from rat | affect | 190 | ||||
| Target | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| mitochondrial KATP chanel (mtKATP) | inhibitor | 188 | ||||||
| mitochondrial KATP chanel (mtKATP) | K1/2 values 1-6 microM | heart and liver mitochondria from rat | inhibitor | 190 | ||||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
 |
Warning |
Aggregated GHS information provided by 220 companies from 10 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. Reported as not meeting GHS hazard criteria by 1 of 220 companies. For more detailed information, please visit ECHA C&L website Of the 9 notification(s) provided by 219 of 220 companies with hazard statement code(s): H302 (96.35%): Harmful if swallowed [Warning Acute toxicity, oral] H413 (97.72%): May cause long lasting harmful effects to aquatic life [Hazardous to the aquatic environment, long-term hazard] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P264, P270, P273, P301+P312, P330, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | subcutaneous | > 20gm/kg (20000mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 19, Pg. 1413, 1969. | |
| dog | LD50 | oral | > 10gm/kg (10000mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 19, Pg. 1404, 1969. | |
| mouse | LD50 | subcutaneous | > 20gm/kg (20000mg/kg) | skin and appendages (skin): "dermatitis, other: after systemic exposure" | Oyo Yakuri. Pharmacometrics. Vol. 4, Pg. 247, 1970. |
| mouse | LD50 | intraperitoneal | 2700mg/kg (2700mg/kg) | Pharmaceutical Chemistry Journal Vol. 29, Pg. 265, 1995. | |
| rat | LD50 | intraperitoneal | 3750mg/kg (3750mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 19, Pg. 1413, 1969. | |
| rabbit | LD | oral | > 20gm/kg (20000mg/kg) | Oyo Yakuri. Pharmacometrics. Vol. 4, Pg. 247, 1970. | |
| rat | LD50 | oral | > 20gm/kg (20000mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 19, Pg. 1413, 1969. | |
| mouse | LD50 | oral | 3250mg/kg (3250mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 16, Pg. 1640, 1966. | |
| guinea pig | LD50 | oral | > 15gm/kg (15000mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 19, Pg. 1404, 1969. | |
| women | TDLo | oral | 147ug/kg (0.147mg/kg) | Drug Intelligence and Clinical Pharmacy. Vol. 18, Pg. 142, 1984. | |
| man | TDLo | oral | 209mg/kg/4Y-I (209mg/kg) | Western Journal of Medicine. Vol. 168, Pg. 274, 1998. | |
| (5-chloro-2-methoxyphenyl)-N-[2-(4-{[(cyclohexylamino)carbonylamino]sulfonyl}p henyl)ethyl]carboxamide | 1-((p-(2-(5-Chloro-o-anisamido)ethyl)phenyl)sulfonyl)-3-cyclohexyl urea | 1-((p-(2-(5-Chloro-o-anisamido)ethyl)phenyl)sulfonyl)-3-cyclohexylurea |
| 1-(p-(2-(5-Chloro-2-methoxybenzamido)ethyl)benzenesulfonyl)-3-cyclohexylurea | 1-[[p-[2-(5-Chloro-o-anisamido)ethyl]phenyl]sulfonyl]-3-cyclohexylurea | 1-{4-[2-(5-chloro-2-methoxybenzamido)ethyl]benzenesulfonyl}-3-cyclohexylurea;1-{4-[2-(5-chloro-2-methoxybenzamido)ethyl]benzenesulfonyl}-3-cyclohexylurea |
| 10238-21-8 | 23047-14-5 | 5-Chloro-N-(2-(4-((((cyclohexylamino)carbonyl)amino)sulfonyl)phenyl)ethyl)-2-methoxybenzamide |
| 5-Chloro-N-(2-[4-(([(cyclohexylamino)carbonyl]amino)sulfonyl)phenyl]ethyl)-2-methoxybenzamide # | 5-Chloro-N-(4-(N-(cyclohexylcarbamoyl)sulfamoyl)-phenethyl)-2-methoxybenzamide | 5-Chloro-N-(4-(N-(cyclohexylcarbamoyl)sulfamoyl)phenethyl)-2-methoxybenzamide |
| 5-Chloro-N-[2-[4-[[[(Cylcohexylamino)carbonyl]amino]sulphonyl]phenyl]ethyl]-2-methoxybenzamide | 5-Chloro-N-[2-[4-[[[(cyclohexylamino)carbonyl]-amino]sulfonyl]phenyl]-ethyl]-2-methoxybenzamide | 5-Chloro-N-[4-(3-cyclohexylureidosulfonyl)phenethyl]-2-methoxybenzamide |
| 5-Chloro-N-[4-(cyclohexylureidosulfonyl)phenethyl]-2-methoxybenzamide | 5-chloro-N-(2-{4-[(cyclohexylcarbamoyl)sulfamoyl]phenyl}ethyl)-2-methoxybenzamide | 5-chloro-N-(2-{4-[N-(N-cyclohexylcarbamoyl)sulfamoyl]phenyl}ethyl)-2-methoxybenzamide |
| 5-chloro-N-(4-[N-(cyclohexylcarbamoyl)sulfamoyl]phenethyl)-2-methoxybenzamide | 5-chloro-N-[2-(4-{[(cyclohexylcarbamoyl)amino]sulfonyl}phenyl)ethyl]-2-methoxybenzamide | 5-chloro-N-[2-[4-(cyclohexylcarbamoylsulfamoyl)phenyl]ethyl]-2-methoxybenzamide |
| 5-chloro-N-[2-[4-(cyclohexylcarbamoylsulfamoyl)phenyl]ethyl]-2-methoxybenzamide. | 5-chloro-N-[4-({[(cyclohexylamino)carbonyl]amino}sulfonyl)phenethyl]-2-methoxybenzamide | 78637-EP2270001A1 |
| 78637-EP2270011A1 | 78637-EP2272825A2 | 78637-EP2272841A1 |
| 78637-EP2275414A1 | 78637-EP2287165A2 | 78637-EP2287166A2 |
| 78637-EP2292620A2 | 78637-EP2298750A1 | 78637-EP2298776A1 |
| 78637-EP2298779A1 | 78637-EP2301923A1 | 78637-EP2301936A1 |
| 78637-EP2305648A1 | 78637-EP2308847A1 | A19539 |
| AB00051949 | AB00051949-16 | AB00051949-17 |
| AB00051949_18 | AB00051949_19 | AB0012611 |
| AB2000243 | ACMC-20a66i | AKOS001487495 |
| ANW-58936 | AOB6214 | Abbenclamide |
| Adiab | Amglidia | Apo-Glibenclamide |
| Azuglucon | BCP05327 | BCP9000729 |
| BDBM50012957 | BG0207 | BIDD:GT0239 |
| BPBio1_000344 | BRD-K36927236-001-06-0 | BRD-K36927236-001-17-7 |
| BRN 2230085 | BSPBio_000312 | BSPBio_001351 |
| BSPBio_003053 | Bastiverit | Benclamin |
| Benzamide, 5-chloro-N-(2-(4-((((cyclohexylamino)carbonyl)amino)sulfonyl)phenyl)ethyl)-2-methoxy- | Benzamide, 5-chloro-N-[2-[4-[[[(cyclohexylamino)carbonyl]amino]sulfonyl]phenyl]ethyl]-2-methoxy- | Betanase |
| Betanese 5 | Bio1_000076 | Bio1_000565 |
| Bio1_001054 | Bio2_000071 | Bio2_000551 |
| C-22898 | C07022 | C23H28ClN3O5S |
| CAS-10238-21-8 | CBiol_001790 | CCG-39618 |
| CHEBI:5441 | CHEMBL472 | CPD000058229 |
| CS-1075 | CTK8B7917 | Calabren |
| Certified Reference Material | Cytagon | D00336 |
| DB01016 | DSSTox_CID_17237 | DSSTox_GSID_37237 |
| DSSTox_RID_79313 | DTXSID0037237 | Daonil |
| Debtan | Delmide | Diabeta |
| Diabeta (TN) | Diabiphage | Dibelet |
| DivK1c_000481 | Duraglucon | EINECS 233-570-6 |
| EU-0100499 | Euclamin | Euglucan |
| Euglucon | Euglucon 5 | Euglucon N |
| Euglykon | FT-0601608 | G 0639 |
| G-150 | G0382 | GBM |
| GBN 5 | GIBENCLAMIDE | GP8701 |
| GTPL2414 | Gen-Glybe | Gewaglucon |
| Gilemal | Glamide | Glibadone |
| Gliban | Gliben | Glibenbeta |
| Glibenclamid AL | Glibenclamid Basics | Glibenclamid Fabra |
| Glibenclamid Genericon | Glibenclamid Heumann | Glibenclamid Riker M |
| Glibenclamid Riker M. | Glibenclamid-Cophar | Glibenclamid-Ratiopharm |
| Glibenclamida | Glibenclamida [INN-Spanish] | Glibenclamide |
| Glibenclamide (JP17/INN) | Glibenclamide 1.0 mg/ml in Acetonitrile | Glibenclamide B.P. |
| Glibenclamide [INN] | Glibenclamide for peak identification, European Pharmacopoeia (EP) Reference Standard | Glibenclamide,(S) |
| Glibenclamidum | Glibenclamidum [INN-Latin] | Glibenil |
| Glibens | Glibesyn | Glibet |
| Glibetic | Glibil | Gliboral |
| Glicem | Glidiabet | Glimel |
| Glimide | Glisulin | Glitisol |
| Glubate | Gluben | Glucobene |
| Glucohexal | Glucolon | Glucomid |
| Glucoremed | Glucoven | Glyben |
| Glybenclamide | Glybenclamide, >=99% (HPLC) | Glybenclamide, European Pharmacopoeia (EP) Reference Standard |
| Glybenzcyclamide | Glyburide (Diabeta) | Glyburide (Glibenclamide), Pharmaceutical Secondary Standard |
| Glyburide (USP) | Glyburide (micronized) | Glyburide [USAN:USP] |
| Glyburide [USAN] | Glyburide(Diabeta) | Glyburide, United States Pharmacopeia (USP) Reference Standard |
| Glyburide, meets USP testing specifications | Glycomin | Glycron |
| Glynase | Glynase (TN) | HB 419 |
| HB-419 | HB-420 | HD 419 |
| HMS1361D13 | HMS1568P14 | HMS1791D13 |
| HMS1922L08 | HMS1989D13 | HMS2089L06 |
| HMS2093P04 | HMS2095P14 | HMS3259O12 |
| HMS3261D19 | HMS3267A15 | HMS3402D13 |
| HMS3411F16 | HMS3428D15 | HMS3651E17 |
| HMS3675F16 | HMS3712P14 | HMS501I03 |
| HY-15206 | Hemi-Daonil | Hexaglucon |
| Humedia | IDI1_000481 | IDI1_033821 |
| J10021 | KBio1_000481 | KBio2_000071 |
| KBio2_000730 | KBio2_002639 | KBio2_003298 |
| KBio2_005207 | KBio2_005866 | KBio3_000141 |
| KBio3_000142 | KBio3_002273 | KBioGR_000071 |
| KBioGR_001897 | KBioSS_000071 | KBioSS_000730 |
| KS-5326 | LP00499 | LS-159295 |
| Lederglib | Libanil | Lisaglucon |
| Lopac-G-0639 | Lopac0_000499 | MCULE-2351642942 |
| MFCD00056625 | MLS000069721 | MLS001077262 |
| Maninil | Med-Glionil | Melix |
| Micronase | Micronase (TN) | Micronized glyburide |
| Miglucan | N-(4-(.beta.-(2-Methoxy-5-chlorbenzamido)-aethyl)-benzolsulfonyl)-N'-cyclohexyl-harnstoff | N-(4-(2-(5-Chloro-2-methoxybenzamido)ethyl)phenylsulfonyl)-N'-cyclohexylurea |
| N-(4-(beta-(2-Methoxy-5-chlorbenzamido)-aethyl)-benzolsulfonyl)-N'-cyclohexyl-harnstoff | N-[(4-{2-[(5-chloro-2-methoxyphenyl)carbonylamino]ethyl}phenyl)sulfonyl](cyclo hexylamino)carboxamide | N-p-[2-(5-Chloro-2-methoxybenzamido)ethyl]benzenesulfonyl-N'-cyclohexylurea |
| N1-[4-({[(cyclohexylamino)carbonyl]amino}sulfonyl)phenethyl]-5-chloro-2-methoxybenzamide | NC00566 | NCGC00015467-01 |
| NCGC00015467-02 | NCGC00015467-03 | NCGC00015467-04 |
| NCGC00015467-05 | NCGC00015467-06 | NCGC00015467-07 |
| NCGC00015467-08 | NCGC00015467-09 | NCGC00015467-10 |
| NCGC00015467-11 | NCGC00015467-12 | NCGC00015467-13 |
| NCGC00015467-14 | NCGC00015467-15 | NCGC00015467-16 |
| NCGC00015467-17 | NCGC00015467-18 | NCGC00015467-20 |
| NCGC00015467-36 | NCGC00016689-01 | NCGC00023447-02 |
| NCGC00023447-04 | NCGC00023447-05 | NCGC00023447-06 |
| NCGC00023447-07 | NCGC00023447-08 | NCGC00023447-09 |
| NCGC00023447-10 | NCGC00023447-11 | NCGC00023447-12 |
| NCGC00254662-01 | NCGC00261184-01 | NINDS_000481 |
| NSC-759618 | NSC759618 | Nadib |
| Neogluconin | Norglicem 5 | Normoglucon |
| Novo-Glyburide | Opera_ID_801 | Oprea1_764617 |
| Orabetic | Pharmakon1600-02300229 | Pira |
| Praeciglucon | Prestwick0_000316 | Prestwick1_000316 |
| Prestwick2_000316 | Prestwick3_000316 | Prestwick_569 |
| Probes1_000431 | Probes2_000378 | Prodiabet |
| Q420626 | RP-1127 | RT-000122 |
| Renabetic | S1716 | SAM002564212 |
| SB17414 | SBB057426 | SBI-0050483.P003 |
| SC-17437 | SCHEMBL22009 | SMR000058229 |
| SPBio_001831 | SPBio_002531 | SPECTRUM2300229 |
| SR-01000000196 | SR-01000000196-2 | SR-01000000196-4 |
| SR-01000000196-5 | SR-01000000196-6 | SR-01000000196-8 |
| ST024780 | STK362992 | SW195828-5 |
| SX6K58TVWC | SYN3026 | Semi-Euglucon |
| Semi-Gliben-Puren N | Semi-daonil | Spectrum2_001816 |
| Spectrum3_001327 | Spectrum4_001199 | Spectrum5_001631 |
| Spectrum_000250 | Sugril | Suraben |
| Tiabet | Tocris-0911 | Tox21_110158 |
| Tox21_110158_1 | Tox21_300758 | Tox21_500499 |
| U 26452 | U-26,452 | U-26452 |
| UNII-SX6K58TVWC | UPCMLD-DP006 | UPCMLD-DP006:001 |
| UR 606 | Urea, 1-((p-(2-(chloro-o-anisamido)ethyl)phenyl)sulfonyl)-3-cyclohexyl- | Urea, 1-(p-(2-(5-chloro-2-methoxybenzamido)ethyl)benzenesulfonyl)-3-cyclohexyl- |
| W-108874 | W-5054 | Yuglucon |
| Z277540138 | ZINC537805 | ZNNLBTZKUZBEKO-UHFFFAOYSA-N |
| glyburide | glyburide (glibenclamide) |