D1332 | buprenorphine
N
N07BC51 Buprenorphine, combinations
[N07BC] Drugs used in opioid dependence
[N07B] DRUGS USED IN ADDICTIVE DISORDERS
[N07] OTHER NERVOUS SYSTEM DRUGS
[N] Nervous system
N07BC01 Buprenorphine
[N07BC] Drugs used in opioid dependence
[N07B] DRUGS USED IN ADDICTIVE DISORDERS
[N07] OTHER NERVOUS SYSTEM DRUGS
[N] Nervous system
N02AE01 Buprenorphine
[N02AE] Oripavine derivatives
[N02A] OPIOIDS
[N02] ANALGESICS
[N] Nervous system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| ELECTROPHORETIC UNCOUPLING | 278 | |||||||
| MITOCHONDRIAL FATTY ACID BETA OXIDATION | 100 μM | rat | liver mitochondria | the mitochondrial membrane potential assessed by the fluorescence of safranine. | affect | 235 | ||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
   |
Danger |
Aggregated GHS information provided by 90 companies from 5 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H301 (87.78%): Toxic if swallowed [Danger Acute toxicity, oral] H302 (11.11%): Harmful if swallowed [Warning Acute toxicity, oral] H315 (87.78%): Causes skin irritation [Warning Skin corrosion/irritation] H319 (90%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H335 (87.78%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure Respiratory tract irritation] H361 (11.11%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P261, P264, P270, P271, P280, P281, P301+P310, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
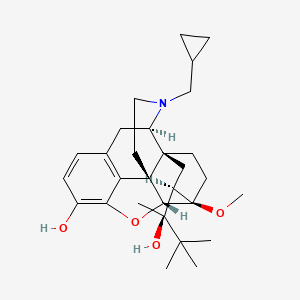
| (-)-buprenorphine | (1S,2R,6S,14R,15R,16R)-3-(cyclopropylmethyl)-16-[(2S)-2-hydroxy-3,3-dimethylbutan-2-yl]-15-methoxy-13-oxa-3-azahexacyclo[13.2.2.1^{2,8}.0^{1,6}.0^{6,14}.0^{7,12}]icosa-7(12),8,10-trien-11-ol | (1S,2R,6S,14R,15R,16R)-3-(cyclopropylmethyl)-16-[(2S)-2-hydroxy-3,3-dimethylbutan-2-yl]-15-methoxy-13-oxa-3-azahexacyclo[13.2.2.1^{2,8}.0^{1,6}.0^{6,14}.0^{7,12}]icosa-7,9,11-trien-11-ol |
| (2S)-2-[(5R,6R,7R,14S)-9alpha-Cyclopropylmethyl-3-hydroxy-6-methoxy-4,5-epoxy-6,14-ethanomorphinan-7-yl]-3,3-dimethylbutan-2-ol | (5alpha,14beta,18R)-17-(Cyclopropylmethyl)-18-[(2S)-2-hydroxy-3,3-dimethyl-2-butanyl]-6-methoxy-18,19-dihydro-4,5-epoxy-6,14-ethenomorphinan-3-ol | (5alpha,6beta,14beta,18R)-17-(cyclopropylmethyl)-18-[(2S)-2-hydroxy-3,3-dimethylbutan-2-yl]-6-methoxy-18,19-dihydro-4,5-epoxy-6,14-ethenomorphinan-3-ol |
| 17-Cyclopropylmethyl-4,5alpha-epoxy-7alpha-((S)-1-hydroxy-1,2,2-trimethylpropyl-6-methoxy-6,14-endo-ethanomorphinan-3-ol | 17-cyclopropylmethyl-4,5alpha-epoxy-7alpha-((S)-1-hydroxy-1,2,2-trimethylpropyl)-6-methoxy-6,14-endo-ethanomorphinan-3-ol | 2-(N-Cyclopropylmethyl-4,5alpha-epoxy-3-hydroxy-6-methoxy-6,14-endo-ethanomorphinan-7alpha-yl)-3,3-dimethyl-2-butanol |
| 2-(N-cyclopropylmethyl-4,5alpha-epoxy-3-hydroxy-6-methoxy-6,14-endo-ethanomorphinan-6alpha-yl)-3,3-dimethyl-2-butanol | 2-[3-cyclopropylmethyl-11-hydroxy-15-methoxy-(14R)-13-oxa-3-azahexacyclo[13.2.2.12,8.01,6.06,14.07,12]icosa-7,9,11-trien-16-yl]-3,3-dimethyl-2-butanol | 21-(Cyclopropyl-7alpha-((S)-1-hydroxy-1,2,2-trimethylpropyl-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine |
| 21-cyclopropyl-7alpha-(2-hydroxy-3,3-dimethyl-2-butyl)-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine | 21-cyclopropyl-7alpha-[(S)-1-hydroxy-1,2,2-trimethylpropyl]-6,14-endo-ethano-6,7,8,14-tetrahydrooripavine | 40D3SCR4GZ |
| 52485-79-7 | 53152-21-9 | 6,14-Ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-, |
| 6,14-Ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-, (5-alpha,7-alpha-(S))- | 6,14-Ethenomorphinan-7-methanol, 17-(cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-, (alphaS,5alpha,7alpha)- (9CI) | 6,14-ethenomorphinan-3-ol, 17-(cyclopropylmethyl)-4,5-epoxy-18,19-dihydro-7-[(1S)-1-hydroxy-1,2,2-trimethylpropyl]-6-methoxy-, (5alpha,7alpha)- |
| 6029-M | BDBM50026603 | BEMA |
| Buprenex | Buprenophine | Buprenorfina |
| Buprenorfina [INN-Spanish] | Buprenorphine (BN) | Buprenorphine (JAN/INN) |
| Buprenorphine [INN:BAN:JAN] | Buprenorphinum | Buprenorphinum [INN-Latin] |
| Butrans | Buvidal | C-22673 |
| C08007 | CAM2038 | CC-25269 |
| CHEBI:3216 | CHEMBL560511 | D07132 |
| DB00921 | DEA No. 9064 | DTXSID2022705 |
| EINECS 257-950-6 | GTPL1670 | Probuphenine |
| Probuphine | Q407721 | RBP-6000 |
| RX 6029M | SCHEMBL15821 | Sixmo (buprenorphine hydrochloride) |
| Sublocade | Subutex | Temgesic |
| Temgesic (TN) | UNII-40D3SCR4GZ | ZINC1319780 |
| [5?,7?(S)]-?-tert-butyl-17-(cyclopropylmethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-?-methyl-6,14-ethenomorphinan-7-methanol hydrochloride;[5a,7a(S)]-a-tert-butyl-17-(cyclopropylmethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-a-methyl-6,14-ethenomorphinan-7-methanol hydrochloride;Buprenorphine hydrochloride | [5alpha,7alpha(S)]- | [5alpha,7alpha(S)]-17-(Cyclopropylmethyl)-alpha-(1,1-dimethylethyl)-4,5-epoxy-18,19-dihydro-3-hydroxy-6-methoxy-alpha-methyl-6,14-ethenomorphinan-7-methanol |
| buprenorphin | buprenorphine |