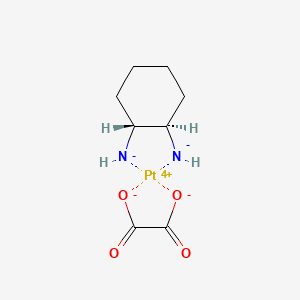
Drug
D1368 | oxaliplatin
L
L01XA03 Oxaliplatin
[L01XA] Platinum compounds
[L01X] OTHER ANTINEOPLASTIC AGENTS
[L01] ANTINEOPLASTIC AGENTS
[L] Antineoplastic and immunomodulating agents
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 30mg/kg (30mg/kg) | Cancer Treatment Reports. Vol. 61, Pg. 1519, 1977. | |
| mouse | LD50 | intraperitoneal | 19800ug/kg (19.8mg/kg) | Rinsho Yakuri. Clinical Pharmacology. Vol. 16, Pg. 147, 1985. | |
| (SP-4-2)-[(1R,2R)-Cyclohexane-1,2-diamine-?N,?N?][ethanedioato(2-)-?O1,?O2]platinum | ACT 078 | CCRIS 9143 |
| Lipoxal | NCI60_002138 | NSC 271670 |
| Oxalato(1,2-diaminocyclohexane)platinum(II) | Oxaliplatin [USAN:USP:INN:BAN] | Oxalitin |
| Oxaloplatine | Oxaloplatino | Platinum (II), (cyclohexane-1,2-diammine)oxalato- |
| Platinum, (1,2-cyclohexanediamine-N,N')(ethanedioato(2-)-O,O')-, (SP-4-2-(trans))- | Platinum, (1,2-cyclohexanediamine-kappaN,kappaN')(ethanedioato(2-)-kappaO1,kappaO2)-, (SP-4-2-(trans))- | [(1R,2R)-Cyclohexane-1,2-diaminato(2-)-kappa~2~N,N'][ethanedioato(2-)-kappa~2~O~1~,O~2~]platinum |