D0019 | Celecoxib
G
L
M
M01AH01 Celecoxib
[M01AH] Coxibs
[M01A] ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS, NON-STEROIDS
[M01] ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS
[M] Musculoskeletal system
L01XX33 Celecoxib
[L01XX] Other antineoplastic agents
[L01X] OTHER ANTINEOPLASTIC AGENTS
[L01] ANTINEOPLASTIC AGENTS
[L] Antineoplastic and immunomodulating agents
G01AE10 Combinations of sulfonamides
[G01AE] Sulfonamides
[G01A] ANTIINFECTIVES AND ANTISEPTICS, EXCL. COMBINATIONS WITH CORTICOSTEROIDS
[G01] GYNECOLOGICAL ANTIINFECTIVES AND ANTISEPTICS
[G] Genitourinary system and reproductive hormones
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| ELONGATION | mouse | neuron | Mitochondrial morphology and neurite area measurements | induce | 269 | |||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
 |
Danger |
Aggregated GHS information provided by 277 companies from 13 notifications to the ECHA C&L Inventory. H360 (92.06%): May damage fertility or the unborn child [Danger Reproductive toxicity] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P281, P308+P313, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
  |
Danger |
H360: May damage fertility or the unborn child [Danger Reproductive toxicity] H401: Toxic to aquatic life [Hazardous to the aquatic environment, acute hazard] H411: Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] |
P201, P202, P273, P281, P308+P313, P391, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| 169590-42-5 | 184007-95-2 | 194044-54-7 |
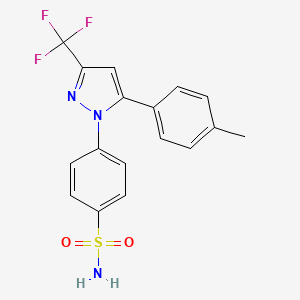
| 1oq5 | 4-(5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl)benzenesulfonamide | 4-(5-(p-tolyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl)benzenesulfonamide |
| 4-[5-(4-METHYLPHENYL)-3-(TRIFLUOROMETHYL)-1H-PYRAZOL-1-YL]BENZENESULFONAMIDE | 4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-pyrazol-1-yl]benzenesulfonamide | 4-[5-(4-Methylphenyl)-3-trifluoromethyl)-1H-pyrazol-yl]benzenesulfonamide |
| 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyazol-1-yl]benezenesulfonamide | 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]-benzenesulfonamide | 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzene-1-sulfonamide |
| 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1Hpyrazol-1-yl] benzenesulfonamide | 4-[5-(4-methylphenyl)-3-(trifluoromethyl)pyrazol-1-yl]benzenesulfonamide | 4-[5-(p-tolyl)-3-(trifluoromethyl)pyrazol-1-yl]benzenesulfonamide |
| 5-(4-Methylphenyl)-1-(4-sulfamoylphenyl)-3-(trifluoromethyl)pyrazole | 590C425 | A25046 |
| AB00052396-07 | AB00052396-08 | AB00052396-09 |
| AB00052396_10 | AB00052396_11 | AB0012055 |
| ABP000291 | AC-4228 | ACT02648 |
| AI-525 | AK-60967 | AKOS015842517 |
| AM84588 | AT-3762 | BBL029086 |
| BCP02156 | BCP9000507 | BCPP000290 |
| BDBM11639 | BIDD:GT0408 | BP-30217 |
| BRD-K02637541-001-02-4 | BRD-K02637541-001-06-5 | BSPBio_003596 |
| Benzenesulfonamide, 4-(5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl)- | Benzenesulfonamide,4-(5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl) | Benzenesulfonamide,4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]- |
| C-6317 | C07589 | C17H14F3N3O2S |
| CAS-169590-42-5 | CCG-39354 | CCRIS 9330 |
| CELEBCOXIB | CEP-33222 | CHEBI:41423 |
| CHEMBL118 | CJ-016377 | CP-598107 |
| CPD000550473 | CS-0570 | CTK8H4475 |
| Celebra | Celebrex | Celebrex (TN) |
| Celebrex, Celebra, 169590-42-5 | Celecox | Celecoxi |
| Celecoxib (JAN/USAN/INN) | Celecoxib (SC-58635) | Celecoxib 1.0 mg/ml in Acetonitrile |
| Celecoxib [USAN:INN:BAN] | Celecoxib [USAN] | Celecoxib, >=98% (HPLC) |
| Celecoxib, European Pharmacopoeia (EP) Reference Standard | Celecoxib, Pharmaceutical Secondary Standard | Celecoxib, United States Pharmacopeia (USP) Reference Standard |
| Celecoxib/Celebrex, Celebra | Celecoxibum | Celocoxib |
| Certified Reference Material | D00567 | DB00482 |
| DSSTox_CID_2777 | DSSTox_GSID_22777 | DSSTox_RID_76725 |
| DTXSID0022777 | DivK1c_000893 | EBD24636 |
| EX-A175 | FT-0080064 | FT-0601628 |
| FT-0623536 | FT-0700357 | GTPL2892 |
| HMS1922G14 | HMS2089L18 | HMS2093I07 |
| HMS2234N18 | HMS3259L08 | HMS3261A14 |
| HMS3373A09 | HMS3654H09 | HMS3715F11 |
| HMS502M15 | HSDB 7038 | HY-14398 |
| IDI1_000893 | J-010566 | J-520011 |
| J10035 | JCX84Q7J1L | KBio1_000893 |
| KBio2_000912 | KBio2_002351 | KBio2_003480 |
| KBio2_004919 | KBio2_006048 | KBio2_007487 |
| KBio3_002830 | KBio3_003037 | KBioGR_000723 |
| KBioGR_002351 | KBioSS_000912 | KBioSS_002354 |
| KS-00000FW2 | KS-1041 | LS-31667 |
| MCULE-4750749400 | MFCD00941298 | MLS001165684 |
| MLS001195656 | MLS001304708 | MLS006011862 |
| NC00708 | NCGC00091455-01 | NCGC00091455-02 |
| NCGC00091455-03 | NCGC00091455-04 | NCGC00091455-05 |
| NCGC00091455-06 | NCGC00091455-07 | NCGC00091455-08 |
| NCGC00091455-09 | NCGC00091455-13 | NCGC00254540-01 |
| NCGC00259513-01 | NCGC00261091-01 | NCI60_041049 |
| NINDS_000893 | NSC-719627 | NSC-758624 |
| NSC719627 | NSC758624 | Onsenal |
| Onsenal (TN) | PF-00345549 | PHA-00846533 |
| Pharmakon1600-01503678 | Q-200816 | Q408801 |
| RZEKVGVHFLEQIL-UHFFFAOYSA-N | SAM002589995 | SB19318 |
| SBI-0051875.P002 | SC 58635 | SC-50829 |
| SC-58553, SC-58635 | SC-58635 | SC58635 |
| SCHEMBL3708 | SMR000550473 | SPBio_001512 |
| SPECTRUM1503678 | SR-01000837528 | SR-01000837528-2 |
| SR-01000837528-3 | STL373576 | SW199611-3 |
| SYN3015 | Solexa | Spectrum2_001576 |
| Spectrum3_001996 | Spectrum4_000182 | Spectrum5_001324 |
| Spectrum_000432 | TPI-336 | Tox21_111135 |
| Tox21_111135_1 | Tox21_201964 | Tox21_300599 |
| Tox21_500406 | UNII-JCX84Q7J1L | UNM-0000305813 |
| US8741944, Comparative Compound | Xilebao | YM 177 |
| YM-177 | YM177 | Z2210694606 |
| ZINC2570895 | benzenesulfonamide, 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]- | cMAP_000027 |
| celecoxib | cid_2662 | p-(5-p-Tolyl-3-(trifluoromethyl)pyrazol-1-yl)benzenesulfonamide |
| s1261 |
| DrugBank Name | Celecoxib |
| DrugBank | DB00482 |
| CAS Number | 1000343-91-8, 169590-42-5, 184007-95-2, 194044-54-7 |
| PubChem Compound | 2662 |
| KEGG Compound ID | C07589 |
| KEGG Drug | D00567 |
| PubChem.Substance | 46505596 |
| ChEBI | 41423 |
| PharmGKB | PA448871 |
| ChemSpider | 2562 |
| BindingDB | 11639.0 |
| TTD | DAP000737 |
| Wikipedia | Celecoxib |
| HET | CEL |
| DPD | 11865 |