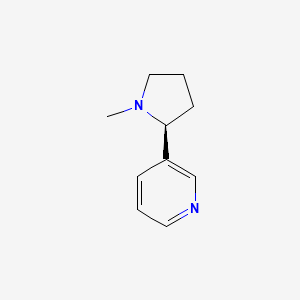
D0091 | Nicotine
N
N07BA01 Nicotine
[N07BA] Drugs used in nicotine dependence
[N07B] DRUGS USED IN ADDICTIVE DISORDERS
[N07] OTHER NERVOUS SYSTEM DRUGS
[N] Nervous system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| MEMBRANE POTENTIAL | > 400 µM | 30 mins | mouse | liver mitochondria | Rh123 fluorescence (excitation 485 nm, emission 535 nm) are recorded using a fluorescence multi-well plate reader (mCICCP (20 µM) treatments was considered as the 100% baseline for ΔΨm loss) | decrease | EC20 | 36 |
| RESPIRATION | > 400 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Rotenone (2µM) was used as 100% baseline for complex I inhibition. | decrease | EC20 | 36 |
| RESPIRATION | 312 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Oligomycin A (1µM) was used as 100% baseline for complex II inhibition. | decrease | EC20 | 36 |
| BASAL RESPIRATION | 1.4 µg/ml | zebrafish | XFe24 Extracellular Flux Analyzer | Negative | 87 | |||
| BASAL RESPIRATION | 1.4 µg/ml | zebrafish | XFe24 Extracellular Flux Analyzer | Negative | 87 | |||
| MAXIMAL RESPIRATION | 1.4 µg/ml | zebrafish | XFe24 Extracellular Flux Analyzer | Negative | 87 | |||
| ATP TURNOVER | 1.4 µg/ml | zebrafish | XFe24 Extracellular Flux Analyzer | Negative | 87 | |||
| SWELLING | > 400 µM | 30 mins | mouse | liver mitochondria | swelling assay: Absorbance at 545 nm using a fluorescence multi-well plate reader (CaCl2 (50 µM) was considered as the 100% baseline for the swelling ) | increase | EC20 | 36 |
| MITOCHONDRIAL DYNAMICS | 10 mM | Human | Human embryonal carcinoma cells (NT2/D1) | 247 | ||||
| Target | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| NADH:ubiquinone reductase | > 400 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Rotenone (2µM) was used as 100% baseline for complex I inhibition. | inhibit | EC20 | 36 |
| Succinate dehydrogenase | 312 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Oligomycin A (1µM) was used as 100% baseline for complex II inhibition. | inhibit | EC20 | 36 |
| Cytochrome c | > 400 µM | 30 mins | mouse | liver mitochondria | Cytochrome c release was evaluated using ELISA kit ( 20 µg/ml Alamethicin was used as 100% baseline) | release | EC20 | 36 |
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
  |
Danger |
H300: Fatal if swallowed [Danger Acute toxicity, oral] H310: Fatal in contact with skin [Danger Acute toxicity, dermal] H330: Fatal if inhaled [Danger Acute toxicity, inhalation] H411: Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] |
P260, P262, P264, P270, P271, P273, P280, P284, P301+P310, P302+P350, P304+P340, P310, P320, P321, P322, P330, P361, P363, P391, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
  |
Danger |
Aggregated GHS information provided by 508 companies from 15 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H300 (69.88%): Fatal if swallowed [Danger Acute toxicity, oral] H301 (30.12%): Toxic if swallowed [Danger Acute toxicity, oral] H310 (99.41%): Fatal in contact with skin [Danger Acute toxicity, dermal] H330 (68.9%): Fatal if inhaled [Danger Acute toxicity, inhalation] H411 (100%): Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P260, P262, P264, P270, P271, P273, P280, P284, P301+P310, P302+P350, P304+P340, P310, P320, P321, P322, P330, P361, P363, P391, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
   |
Danger |
The GHS information provided by 1 company from 1 notification to the ECHA C&L Inventory. H301 (100%): Toxic if swallowed [Danger Acute toxicity, oral] H310 (100%): Fatal in contact with skin [Danger Acute toxicity, dermal] H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H411 (100%): Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] |
P262, P264, P270, P273, P280, P301+P310, P302+P350, P305+P351+P338, P310, P321, P322, P330, P337+P313, P361, P363, P391, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
  |
Danger |
The GHS information provided by 1 company from 1 notification to the ECHA C&L Inventory. H300 (100%): Fatal if swallowed [Danger Acute toxicity, oral] H310 (100%): Fatal in contact with skin [Danger Acute toxicity, dermal] H411 (100%): Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] |
P262, P264, P270, P273, P280, P301+P310, P302+P350, P310, P321, P322, P330, P361, P363, P391, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
  |
Danger |
H300: Fatal if swallowed [Danger Acute toxicity, oral] H310: Fatal in contact with skin [Danger Acute toxicity, dermal] H330: Fatal if inhaled [Danger Acute toxicity, inhalation] H411: Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard] |
P260, P262, P264, P270, P271, P273, P280, P284, P301+P310, P302+P350, P304+P340, P310, P320, P321, P322, P330, P361, P363, P391, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
   |
Danger |
H300: Fatal if swallowed [Danger Acute toxicity, oral] H310: Fatal in contact with skin [Danger Acute toxicity, dermal] H335: May cause respiratory irritation [Warning Specific target organ toxicity, single exposure Respiratory tract irritation] H361: Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] H370: Causes damage to organs [Danger Specific target organ toxicity, single exposure] |
P201, P202, P260, P261, P262, P264, P270, P271, P280, P281, P301+P310, P302+P350, P304+P340, P307+P311, P308+P313, P310, P312, P321, P322, P330, P361, P363, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| bird - domestic | LDLo | intramuscular | 25mg/kg (25mg/kg) | Physiological Zoology. Vol. 12, Pg. 238, 1939. | |
| guinea pig | LDLo | intraperitoneal | 33mg/kg (33mg/kg) | Australian Journal of Experimental Biology and Medical Science. Vol. 25, Pg. 83, 1947. | |
| rat | LDLo | subcutaneous | 50mg/kg (50mg/kg) | "Structure et Activite Pharmacodyanmique des Medicaments du Systeme Nerveux Vegetatif," Bovet, D., and F. Bovet-Nitti, New York, S. Karger, 1948Vol. -, Pg. 584, 1948. | |
| rat | LDLo | intraperitoneal | 24mg/kg (24mg/kg) | Australian Journal of Experimental Biology and Medical Science. Vol. 25, Pg. 83, 1947. | |
| frog | LDLo | unreported | 40mg/kg (40mg/kg) | "Structure et Activite Pharmacodyanmique des Medicaments du Systeme Nerveux Vegetatif," Bovet, D., and F. Bovet-Nitti, New York, S. Karger, 1948Vol. -, Pg. 584, 1948. | |
| mouse | LD50 | intravenous | 6150ug/kg (6.15mg/kg) | Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. Vol. 84, Pg. 1, 1984. | |
| (-)-.beta.-Pyridyl-.alpha.-N-methylpyrrolidine | (-)-1-Methyl-2-(3-pyridyl)pyrrolidine | (-)-3-(1-Methyl-2-pyrrolidyl)pyridine |
| (-)-3-(N-Methylpyrrolidino)pyridine | (-)-Nicotine | (-)-Nicotine solution, 1.0 mg/mL, analytical standard, for drug analysis |
| (-)-Nicotine solution, 100 mug/mL in acetonitrile, PESTANAL(R), analytical standard | (-)-Nicotine, >=99% (GC), liquid | (-)-Nicotine, PESTANAL(R), analytical standard |
| (2S) 3-(1-Methyl-pyrrolidin-2-yl)-pyridine | (S)-(-)--nicotine | (S)-(-)-Nicotine |
| (S)-3-(1-Methyl-2-pyrrolidinyl)pyridine | (S)-3-(1-methylpyrrolidin-2-yl)pyridine | (S)-3-(N-methylpyrrolidin-2-yl)pyridine |
| (S)-Nicotine | 006N369 | 1-Methyl-2-(3-pyridiyl)pyrrolidine |
| 1-Methyl-2-(3-pyridyl)pyrrolidine | 1-methyl-2-(3-pyridal)-Pyrrolidine | 1-methyl-2-(3-pyridal)-pyrrolidene |
| 1uw6 | 3-((2S)-1-methylpyrrolidin-2-yl)pyridine | 3-(1-Methyl-2-pyrollidinyl)pyridine |
| 3-(1-Methyl-2-pyrrolidinyl)pyridine | 3-(1-methyl-2-pyrrolidinyl)-Pyridine | 3-(2-(N-methylpyrrolidinyl))pyridine |
| 3-(N-Methylpyrollidino)pyridine | 3-(N-Methylpyrrolidino)pyridine | 3-N-methylpyrrolidine |
| 3-[(1R)-1beta-Methylpyrrolidine-2alpha-yl]pyridine | 3-[(2S)-1-METHYL-2-PYRROLIDINYL] PYRIDINE | 3-[(2S)-1-methylpyrrolidin-2-yl]pyridine |
| 434F7990-3240-4A43-ACEC-E6CC1E495FA0 | 54-11-5 | 6M3C89ZY6R |
| AB00694322_12 | AI3-03424 | AKOS016843798 |
| BDBM82070 | BIDD:GT0599 | BRD-K05395900-322-02-1 |
| BRD-K05395900-322-04-7 | Black leaf | C00745 |
| CAS-54-11-5 | CAS_29790-52-1 | CCG-204892 |
| CCRIS 1637 | CHEBI:17688 | CHEMBL3 |
| CPD000059074 | CS-3999 | CTK8F2098 |
| Campbell's nico-soap | Caswell No. 597 | D03365 |
| DB00184 | DL-tetrahydronicotyrine | DSSTox_CID_930 |
| DSSTox_GSID_20930 | DSSTox_RID_75874 | DTXSID1020930 |
| Destruxol orchid spray | EC 200-193-3 | EINECS 200-193-3 |
| ENT 3,424 | EPA Pesticide Chemical Code 056702 | Emo-nik |
| Exodus | FT-0603228 | FT-0672702 |
| Flux MAAG | Fumetobac | GTPL2585 |
| HMS2230H17 | HMS3259E16 | HSDB 1107 |
| HY-B0638 | Habitrol | Habitrol (TN) |
| J-500021 | L(-)-nicotine | L-3-(1-Methyl-2-pyrrolidyl)pyridine |
| L-Nicotine | L-Nicotine, 99+% | MCULE-8728421654 |
| MFCD00006369 | MLS001055457 | MLS001335905 |
| Mach-Nic | Micotine | N0079 |
| NC00577 | NCGC00090693-01 | NCGC00090693-02 |
| NCGC00090693-03 | NCGC00090693-04 | NCGC00090693-05 |
| NCGC00090693-06 | NCGC00090693-07 | NCGC00090693-08 |
| NCGC00090693-09 | NCGC00254095-01 | NCGC00259363-01 |
| NICOTINE AND SALTS | NICOTINE-L (BASE) | NSC 5065 |
| Niagara P.A. dust | Nic-Sal | Nicabate |
| Nico-Fume | Nico-dust | Nicocide |
| Nicoderm | Nicoderm Cq | Nicoderm Patch |
| Nicorette | Nicotin | Nicotina |
| Nicotina [Italian] | Nicotine (USP) | Nicotine (compounds related to) |
| Nicotine 10 microg/mL in Methanol | Nicotine 100 microg/mL in Methanol | Nicotine Patch |
| Nicotine [BSI:ISO] | Nicotine [UN1654] | Nicotine [UN1654] [Poison] |
| Nicotine [UN1654] [Poison] | Nicotine [USAN] | Nicotine [USP:BAN] |
| Nicotine [for single use] | Nicotine alkaloid | Nicotine betadex |
| Nicotine polacrilex | Nicotine polacrilex [USAN] | Nicotine-D salicylate |
| Nicotinum | Nicotrol | Nicotrol Inhaler |
| Nicotrol NS | Nictoine patch | Nikotin [German] |
| Nikotyna | Nikotyna [Polish] | Niquitin |
| Ortho N-4 and N-5 dusts | Ortho N-4 dust | Ortho N-5 dust |
| PDSP1_000113 | PDSP1_000465 | PDSP2_000463 |
| PDSP2_000555 | Prostep | Pyridine, 3-((2S)-1-methyl-2-pyrrolidinyl)- |
| Pyridine, 3-(1-methyl-2-pyrrolidinyl)-, (S)- | Pyridine, 3-(1-methyl-2-pyrrolidinyl)-, (S)- (9CI) | Pyridine, 3-(tetrahydro-1-methylpyrrol-2-yl) |
| Pyridine, 3-(tetrahydro-1-methylpyrrol-2-yl), (S)- | Pyridine, 3-[(2S)-1-methyl-2-pyrrolidinyl]- | Pyrrolidine, 1-methyl-2-(3-pyridal)- |
| Pyrrolidine, 1-methyl-2-(3-pyridyl)- | Q28086552 | R)-(+)-nicotine |
| RCRA waste no. P075 | RCRA waste number P075 | S(-)-Nicotine solution, 1.0 mg/mL in methanol, ampule of 1 mL, certified reference material |
| S-()-Nicotine-pyridine-d4 | S-(-)-Nicotine | SAM002564224 |
| SB12751 | SBB012359 | SBI-0050785.P003 |
| SC-58411 | SCHEMBL20192 | SDCCGMLS-0066911.P001 |
| SMR000059074 | SNICXCGAKADSCV-JTQLQIEISA-N | SR-05000001762-5 |
| ST069320 | Tabazur | Tendust |
| Tetrahydronicotyrine, DL- | Tox21_201814 | Tox21_300174 |
| Transdermal Nicotine | UN1654 | UNII-6M3C89ZY6R |
| XL All Insecticide | Z1954805269 | ZINC391812 |
| a -N-methylpyrrolidine | a-N-methylpyrrolidine | alpha-N-methylpyrrolidine |
| beta-Pyridyl-alpha-N-methyl pyrrolidine | beta-Pyridyl-alpha-N-methylpyrrolidine | bmse000105 |
| delta-Nicotine | destruxol | fumeto bac |
| methyl-2-pyrrolidinyl)pyridine | nicotine | nicotine replacement patch |
| DrugBank Name | Nicotine |
| DrugBank | DB00184 |
| CAS Number | 104062-50-2, 25013-16-5, 25162-00-9, 54-11-5, 65-31-6, 69782-38-3 |
| PubChem Compound | 89594 |
| KEGG Compound ID | C00745 |
| KEGG Drug | D03365 |
| PubChem.Substance | 46506924 |
| ChEBI | 17688 |
| PharmGKB | PA450626 |
| ChemSpider | 80863 |
| BindingDB | 82070.0 |
| TTD | DAP000175 |
| Wikipedia | Nicotine |
| HET | NCT |
| DPD | 4365 |