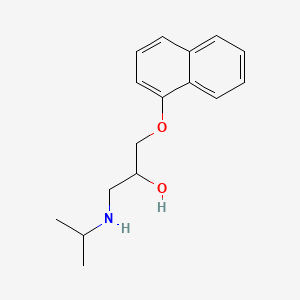
D0502 | propranolol
C
C07FX01 Propranolol and other combinations
[C07FX] Beta blocking agents, other combinations
[C07F] BETA BLOCKING AGENTS, OTHER COMBINATIONS
[C07] BETA BLOCKING AGENTS
[C] Cardiovascular system
C07BA05 Propranolol and thiazides
[C07BA] Beta blocking agents, non-selective, and thiazides
[C07B] BETA BLOCKING AGENTS AND THIAZIDES
[C07] BETA BLOCKING AGENTS
[C] Cardiovascular system
C07AA05 Propranolol
[C07AA] Beta blocking agents, non-selective
[C07A] BETA BLOCKING AGENTS
[C07] BETA BLOCKING AGENTS
[C] Cardiovascular system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| UNCOUPLING | rat | isolated liver mitochondria | measurements of mitochondrial respiration; RST inhibition assay, RST uncoupling assay; IC 50ratio of glucose/galactose assay | Negative | 53 | |||
| ELECTROPHORETIC UNCOUPLING | 278 | |||||||
| ELECTRON TRANSPORT CHAIN | pig | brain mitochondria | decrease | 60 | ||||
| ELECTRON TRANSPORT CHAIN | rat | isolated liver mitochondria | measurements of mitochondrial respiration; RST inhibition assay, RST uncoupling assay; IC 50ratio of glucose/galactose assay | Negative | 53 | |||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
 |
Warning |
Aggregated GHS information provided by 2 companies from 1 notifications to the ECHA C&L Inventory. H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P264, P270, P301+P312, P330, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| man | LDLo | oral | 137mg/kg (137mg/kg) | Journal of Forensic Sciences. Vol. 27, Pg. 213, 1982. | |
| women | LDLo | oral | 120mg/kg (120mg/kg) | Acta Pharmacologica et Toxicologica. Vol. 41, Pg. 190, 1977. | |
| human | TDLo | oral | 2300ug/kg/D (2.3mg/kg) | behavioral: "hallucinations, distorted perceptions" | British Medical Journal. Vol. 1, Pg. 1182, 1978. |
| cat | LD50 | intravenous | 23mg/kg (23mg/kg) | Polish Journal of Pharmacology and Pharmacy. Vol. 32, Pg. 833, 1980. | |
| mouse | LD50 | subcutaneous | 150mg/kg (150mg/kg) | Farmakologiya i Toksikologiya Vol. 44, Pg. 342, 1981. | |
| man | TDLo | oral | 8343mg/kg/4Y- (8343mg/kg) | endocrine: evidence of thyroid hyperfunction | Archives of Internal Medicine. Vol. 143, Pg. 2193, 1983. |
| women | TDLo | oral | 708mg/kg/39W- (708mg/kg) | Annals of Pharmacotherpy. Vol. 27, Pg. 512, 1993. | |
| human | LDLo | intravenous | 71ug/kg (0.071mg/kg) | Lancet. Vol. 1, Pg. 165, 1967. | |
| rat | LD50 | intravenous | 2750ug/kg (2.75mg/kg) | Polish Journal of Pharmacology and Pharmacy. Vol. 32, Pg. 823, 1980. | |
| guinea pig | LD50 | intravenous | 26mg/kg (26mg/kg) | Dissertationes Pharmaceuticae et Pharmacologicae. Vol. 24, Pg. 103, 1972. | |
| man | TDLo | oral | 45714ug/kg (45.714mg/kg) | Pediatric Emergency Care. Vol. 15, Pg. 262, 1999. | |
| rat | LD50 | intravenous | 23mg/kg (23mg/kg) | Pharmazie. Vol. 31, Pg. 635, 1976. | |
| child | LDLo | oral | 800ug/kg/12H (0.8mg/kg) | British Medical Journal. Vol. 2, Pg. 254, 1978. | |
| mouse | LD50 | oral | 289mg/kg (289mg/kg) | autonomic nervous system: beta adrenergic blockage | Arzneimittel-Forschung. Drug Research. Vol. 30, Pg. 1831, 1980. |
| women | TDLo | oral | 3200ug/kg/2D- (3.2mg/kg) | endocrine: hypoglycemia | Israel Journal of Medical Sciences. Vol. 18, Pg. 725, 1982. |
| rabbit | LD50 | intravenous | 18500ug/kg (18.5mg/kg) | Polish Journal of Pharmacology and Pharmacy. Vol. 32, Pg. 833, 1980. | |
| man | TDLo | unreported | 22857ug/kg/40 (22.857mg/kg) | sense organs and special senses: change in acuity: ear | British Medical Journal. Vol. 289, Pg. 1490, 1984. |
| dog | LDLo | oral | 120mg/kg (120mg/kg) | Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. Vol. 69(4), Pg. 262P, 1973. | |
| child | TDLo | oral | 400mg/kg (400mg/kg) | Medical Journal of Australia. Vol. 1, Pg. 82, 1981. | |
| mouse | LD50 | intravenous | 24400ug/kg (24.4mg/kg) | United States Patent Document. Vol. #4013663, | |
| rat | LD50 | oral | 660mg/kg (660mg/kg) | Pharmazie. Vol. 31, Pg. 635, 1976. | |
| mouse | LD50 | intraperitoneal | 42mg/kg (42mg/kg) | autonomic nervous system: beta adrenergic blockage | Ain Shams Medical Journal. Vol. 30, Pg. 23, 1979. |
| dog | LDLo | intraperitoneal | 40mg/kg (40mg/kg) | behavioral: convulsions or effect on seizure threshold | Pharmazie. Vol. 31, Pg. 635, 1976. |
| (+-)-Propranolol | (+/-)-Propranolol-D7 (ring-D7) solution, 100 mug/mL in methanol with 5% 1 M HCl, ampule of 1 mL, certified reference material; | (.+/-.)-Propranolol |
| (1)-1-(Isopropylamino)-3-(naphthyloxy)propan-2-ol | (A+/-)-Propranolol hydrochloride | .beta.-Propranolol |
| 1(-alpha-naphthoxy)-3-(iso-propylamino)-2-propanol | 1-((1-Methylethyl)amino)-3-(1-naphthalenyloxy)-2-propanol | 1-((1-Methylethyl)amino)-3-(1-naphthyloxy)-2-propanol |
| 1-(1-Naphthyloxy)-2-hydroxy-3-(isopropylamino)propane | 1-(Isopropylamino)-3-(1-naphthoxy)-propan-2-ol | 1-(Isopropylamino)-3-(naphthalen-1-yloxy)propan-2-ol |
| 1-(alpha-naphthoxy)-3-(iso-propylamino)-2-propanol | 1-(alpha-naphthoxy)-3-(isopropylamino)-2-propanol | 1-(isopropylamino)-3-(1-naphthyloxy)propan-2-ol |
| 1-(isopropylamino)-3-naphthalen-1-yloxy-propan-2-ol | 1-(naphthalen-1-yloxy)-3-(propan-2-ylamino)propan-2-ol | 1-Isopropylamino-3-(1-naphthyloxy)-2-propanol |
| 1-[(1-methylethyl)amino]-3-(naphthalen-1-yloxy)propan-2-ol | 1-naphthalen-1-yloxy-3-(propan-2-ylamino)propan-2-ol | 13013-17-7 |
| 2-Propanol, 1-((1-methylethyl)amino)-3-(1-naphthalenyloxy)- | 2-Propanol, 1-((1-methylethyl)amino)-3-(1-naphthalenyloxy)- (9CI) | 2-Propanol, 1-((1-methylethyl)amino)-3-(1-naphthalenyloxy)-, (+-)- |
| 2-Propanol, 1-((1-methylethyl)amino)-3-(1-naphthalenyloxy)-, (+-)- (9CI) | 2-Propanol, 1-(isopropylamino)-3-(1-naphthyloxy)- | 2-Propanol, 1-(isopropylamino)-3-(1-naphthyloxy)-, (+-)- |
| 2-Propanol, 1-[(1-methylethyl)amino]-3-(1-naphthalenyloxy)- | 3-(naphthalen-1-yloxy)-1-(propan-2-ylamino)propan-2-ol | 3-[(methylethyl)amino]-1-naphthyloxypropan-2-ol |
| 525-66-6 | 525P666 | AB00053537 |
| AB00053537-10 | AB00053537_11 | AB00053537_12 |
| AB0108847 | AKOS000588816 | AKOS016050338 |
| ALBB-023324 | AQHHHDLHHXJYJD-UHFFFAOYSA-N | ARONIS27011 |
| AY 20694 | AY-20694 | AY-64043- |
| Anapriline | Avlocardyl | Avlocardyl (Salt/Mix) |
| BBL023437 | BCP26001 | BDBM25761 |
| BPBio1_001040 | BRD-A10070317-003-06-9 | BRD-A10070317-003-17-6 |
| BSPBio_000944 | BSPBio_002682 | Bedranol |
| Betadren | Betalong | Bio1_000367 |
| Bio1_000856 | Bio1_001345 | C07407 |
| CBDivE_006180 | CCG-103643 | CCRIS 3082 |
| CHEBI:8499 | CHEMBL27 | CS-0069968 |
| CTK8I6896 | Corpendol | D,L-Propranolol |
| D08443 | DB00571 | DTXSID6023525 |
| DivK1c_000023 | Dociton | Dociton (Salt/Mix) |
| EINECS 208-378-0 | EINECS 235-867-6 | EN300-40731 |
| Euprovasin | F0001-3681 | FE-0204 |
| FT-0700657 | GTPL564 | HMS2090L21 |
| HMS3428G03 | HY-B0573B | ICI 45520 (Salt/Mix) |
| IDI1_000023 | Inderal | Inderal (Salt/Mix) |
| Inderal hydrochloride | Inderol | KBio1_000023 |
| KBio2_002515 | KBio2_005083 | KBio2_007651 |
| KBio3_001766 | KBio3_001902 | KBio3_002993 |
| KBioGR_001347 | KBioGR_001684 | KBioGR_002515 |
| KBioSS_002523 | KS-00002WT3 | L000679 |
| LS-122410 | LS-184129 | Lopac0_000896 |
| MCULE-7804628650 | NCGC00015798-04 | NCGC00015798-05 |
| NCGC00015798-06 | NCGC00015798-07 | NCGC00015798-08 |
| NCGC00015798-09 | NCGC00024690-02 | NCGC00024690-03 |
| NINDS_000023 | NSC 91523 (Salt/Mix) | NSC91523 |
| Obsidan (Salt/Mix) | Obzidan | Oprea1_304193 |
| PDSP1_000767 | PDSP1_001607 | PDSP1_001608 |
| PDSP2_000755 | PDSP2_001591 | PDSP2_001592 |
| PROPANOLOL(-) | PROPRANOLOL, d | PROPRANOLOL,(+) |
| PROPRANOLOL,(-) | Pranolol | Prestwick0_000952 |
| Prestwick1_000952 | Prestwick2_000952 | Prestwick3_000952 |
| Propanalol | Propanix | Propanolol |
| Propanolol [INN-Spanish] | Propanolol,(+/-) | Propranalol |
| Propranolol (INN) | Propranolol (TN) | Propranolol [INN:BAN] |
| Propranololo [DCIT] | Propranololum | Propranololum [INN-Latin] |
| Proprasylyt | Q423364 | Racemic propranolol |
| Reducor | SBI-0050871.P003 | SCHEMBL12264958 |
| SCHEMBL3955 | SPBio_001361 | SPBio_001658 |
| SPBio_003093 | ST024757 | STK735510 |
| Sawatal | Spectrum2_001301 | Spectrum2_001699 |
| Spectrum3_000883 | Spectrum3_001071 | Spectrum4_000974 |
| Spectrum4_001222 | Spectrum5_000751 | Sumial |
| T7896 | W-109550 | [2-hydroxy-3-(naphthalen-1-yloxy)propyl](propan-2-yl)amine |
| b-Propranolol | beta-Propranolol | cMAP_000071 |
| dl-propranolol | hemangiol | propranolol |
| propranololo | racemic-Propranolol |