D0944 | fluticasone propionate
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| MEMBRANE POTENTIAL | 9.02±1.71 | human | qHTS-HepG2 | MMP assay | decrease | IC50 | 163 | |
| MEMBRANE POTENTIAL | human | HepG2 | MMP assay | Negative | IC50 | 163 | ||
| MEMBRANE POTENTIAL | rat | hepatocytes | MMP assay | Negative | IC50 | 163 | ||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
   |
Danger |
Aggregated GHS information provided by 15 companies from 13 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. Reported as not meeting GHS hazard criteria by 1 of 15 companies. For more detailed information, please visit ECHA C&L website Of the 12 notification(s) provided by 14 of 15 companies with hazard statement code(s): H302 (14.29%): Harmful if swallowed [Warning Acute toxicity, oral] H317 (14.29%): May cause an allergic skin reaction [Warning Sensitization, Skin] H334 (14.29%): May cause allergy or asthma symptoms or breathing difficulties if inhaled [Danger Sensitization, respiratory] H360 (42.86%): May damage fertility or the unborn child [Danger Reproductive toxicity] H361 (50%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] H373 (64.29%): Causes damage to organs through prolonged or repeated exposure [Warning Specific target organ toxicity, repeated exposure] H410 (14.29%): Very toxic to aquatic life with long lasting effects [Warning Hazardous to the aquatic environment, long-term hazard] H413 (14.29%): May cause long lasting harmful effects to aquatic life [Hazardous to the aquatic environment, long-term hazard] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P260, P261, P264, P270, P272, P273, P280, P281, P285, P301+P312, P302+P352, P304+P341, P308+P313, P314, P321, P330, P333+P313, P342+P311, P363, P391, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 980mg/kg (980mg/kg) | Journal of the American Pharmaceutical Association, Scientific Edition. Vol. 47, Pg. 479, 1958. | |
| cat | LDLo | intraperitoneal | 1gm/kg (1000mg/kg) | lungs, thorax, or respiration: other changes | Journal of Pharmacology and Experimental Therapeutics. Vol. 101, Pg. 119, 1951. |
| dog | LDLo | intraperitoneal | 1gm/kg (1000mg/kg) | lungs, thorax, or respiration: other changes | Journal of Pharmacology and Experimental Therapeutics. Vol. 101, Pg. 119, 1951. |
| mouse | LD50 | oral | 300mg/kg (300mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 5, Pg. 572, 1955. | |
| rabbit | LD50 | oral | 3200mg/kg (3200mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 5, Pg. 572, 1955. | |
| rabbit | LDLo | intraperitoneal | 1gm/kg (1000mg/kg) | lungs, thorax, or respiration: other changes | Journal of Pharmacology and Experimental Therapeutics. Vol. 101, Pg. 119, 1951. |
| cat | LDLo | oral | 150mg/kg (150mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 5, Pg. 572, 1955. | |
| mouse | LD50 | intravenous | 313mg/kg (313mg/kg) | behavioral: general anesthetic | Journal of Pharmacology and Experimental Therapeutics. Vol. 101, Pg. 119, 1951. |
| rat | LD50 | intraperitoneal | 600mg/kg (600mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 108, Pg. 450, 1953. | |
| guinea pig | LD50 | oral | 1730mg/kg (1730mg/kg) | Journal of the American Pharmaceutical Association, Scientific Edition. Vol. 47, Pg. 479, 1958. | |
| mouse | LD50 | intraperitoneal | 180mg/kg (180mg/kg) | Bollettino Chimico Farmaceutico. Vol. 111, Pg. 293, 1972. | |
| guinea pig | LDLo | intraperitoneal | 1gm/kg (1000mg/kg) | lungs, thorax, or respiration: other changes | Journal of Pharmacology and Experimental Therapeutics. Vol. 101, Pg. 119, 1951. |
| mouse | LD50 | subcutaneous | 300mg/kg (300mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 8, Pg. 25, 1958. | |
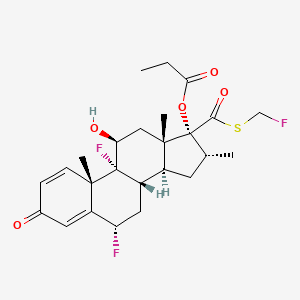
| (1R,2S,8S,10S,11S,13R,14R,15S,17S)-1,8-difluoro-14-{[(fluoromethyl)sulfanyl]carbonyl}-17-hydroxy-2,13,15-trimethyl-5-oxotetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-3,6-dien-14-yl propanoate | (6?,11?,16?,17?)-6,9-Difluoro-11-hydroxy-16-methyl-3-oxo-17-(1-oxopropoxy)androsta-1,4-diene-17-carbothioic acid fluoromethyl ester | (6alpha,11beta,16alpha,17alpha)-6,9-Difluoro-11-hydroxy-16-methyl-3-oxo-17-(1-oxopropoxy)androsta-1,4-diene-17-carbothioic acid S-(fluoromethyl) ester |
| 474F142 | 6alpha,9-difluoro-17beta-{[(fluoromethyl)sulfanyl]carbonyl}-11beta-hydroxy-16-methyl-3-oxoandrosta-1,4-dien-17alpha-yl propanoate | 80474-14-2 |
| AB00513992 | AB00513992-06 | AB00513992_08 |
| AB1010912 | AC-457 | AKOS015895220 |
| AN-584/43505443 | Androsta-1,4-diene-17-carbothioic acid, 6,9-difluoro-11-hydroxy-16-methyl-3-oxo-17-(1-oxopropoxy)-, (6alpha,11beta,16alpha,17alpha)-S-(fluoromethyl) ester | Androsta-1,4-diene-17-carbothioic acid, 6,9-difluoro-11-hydroxy-16-methyl-3-oxo-17-(1-oxopropoxy)-, S-(fluoromethyl) ester, (6-alpha,11-beta,16-alpha,17-alpha)- |
| ArmonAir RespiClick | ArmonAir RespiClickTM | Asmatil |
| Axotide | BDBM50354849 | BIF0710 |
| BPBio1_001203 | BR-72799 | BRD-K62310379-001-03-0 |
| BSPBio_001093 | Brethal | C25H31F3O5S |
| CCG-100981 | CCI 18781 | CCI-18781 |
| CHEBI:31441 | CHEMBL1473 | CS-1986 |
| Certified Reference Material | Cultivate | Cutivate |
| Cutivate (TN) | D01708 | DB00588 |
| FLUTICASONE PROPIONATE | FN-25 | FT-0082893 |
| Flixonase | Flixonase Nasal Spray | Flixotide |
| Flixotide Disk | Flixotide Disks | Flixotide Inhaler |
| Flonase | Flonase (TN) | Flonase Allergy Relief |
| Flonase Aq | Flovent | Flovent (TN) |
| Flovent Diskus | Flovent Diskus 100 | Flovent Diskus 250 |
| Flovent Diskus 50 | Flovent HFA | Flovent Rotadisk |
| Flovent-hfa | Fluinol | Flunase |
| Flusonal | Fluspiral | Fluticasone (propionate) |
| Fluticasone Impurity B | Fluticasone Propionate, Pharmaceutical Secondary Standard | Fluticasone propionate (Flonase, Veramyst) |
| Fluticasone propionate (JAN/USAN) | Fluticasone propionate [USAN:USP] | Fluticasone propionate [USAN] |
| Fluticasone propionate for impurity C identification, EuropePharmacopoeia (EP) Reference Standard | Fluticasone propionate for impurity G identification, EuropePharmacopoeia (EP) Reference Standard; | Fluticasone propionate, >=98% (HPLC), powder |
| Fluticasone propionate, European Pharmacopoeia (EP) Reference Standard | Fluticasone propionate, United States Pharmacopeia (USP) Reference Standard | Fluticasonpropionat Allen |
| Flutide | Flutivate | Fluxonal |
| GTPL7080 | HMS1571G15 | HMS2051N19 |
| HMS2098G15 | HMS3413A19 | HMS3677A19 |
| HMS3715G15 | HY-B0154 | Inalacor |
| KS-1173 | LS-19323 | MFCD00866007 |
| MLS001424085 | NC00231 | NCGC00179308-01 |
| NCGC00179308-05 | O2GMZ0LF5W | PF-00241939 |
| Prestwick0_000997 | Prestwick1_000997 | Prestwick2_000997 |
| Prestwick3_000997 | Q-101393 | Q8564098 |
| Rinosone | S-(Fluoromethyl) 6alpha,9-difluoro-11beta,17-dihydroxy-16alpha-methyl-3-oxoandrosta-1,4-diene-17beta-carbothioate, 17-propionate | SC-19333 |
| SCHEMBL4068 | SMR000469159 | SPBio_002984 |
| SR-01000763355 | SR-01000763355-3 | ST2407016 |
| Skyron | Trialona | UNII-O2GMZ0LF5W |
| Ubizol | WMWTYOKRWGGJOA-CENSZEJFSA-N | Xhance |
| ZINC3920027 | Zoflut | [(6S,8S,9R,10S,11S,13S,14S,16R,17R)-6,9-difluoro-17-(fluoromethylsulfanylcarbonyl)-11-hydroxy-10,13,16-trimethyl-3-oxo-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-17-yl] propanoate |
| atemur | s1992 |