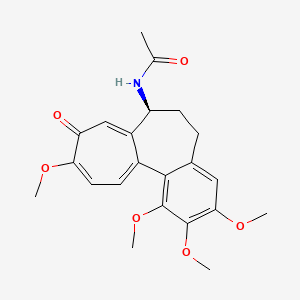
D1372 | colchicine
M
M04AC01 Colchicine
[M04AC] Preparations with no effect on uric acid metabolism
[M04A] ANTIGOUT PREPARATIONS
[M04] ANTIGOUT PREPARATIONS
[M] Musculoskeletal system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| GLUCOSE GALACTOSE IC50 RATIO | 0.5 | LUHMES (Lund human mesencephalic) cells | Glc–Gal–NeuriTox assay | Negative | EC25(NA) [Glc/Gal] | 326 | ||
| FUSION | 185 | |||||||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
  |
Danger |
H300: Fatal if swallowed [Danger Acute toxicity, oral] H340: May cause genetic defects [Danger Germ cell mutagenicity] |
P201, P202, P264, P270, P281, P301+P310, P308+P313, P321, P330, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
   |
Danger |
Aggregated GHS information provided by 272 companies from 7 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H300+H330 (13.97%): Fatal if swallowed or if inhaled [Danger Acute toxicity, oral acute toxicity, inhalation] H300 (80.51%): Fatal if swallowed [Danger Acute toxicity, oral] H310 (19.12%): Fatal in contact with skin [Danger Acute toxicity, dermal] H318 (13.97%): Causes serious eye damage [Danger Serious eye damage/eye irritation] H330 (14.34%): Fatal if inhaled [Danger Acute toxicity, inhalation] H340 (98.9%): May cause genetic defects [Danger Germ cell mutagenicity] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P260, P262, P264, P270, P271, P280, P281, P284, P301+P310, P302+P350, P304+P340, P305+P351+P338, P308+P313, P310, P320, P321, P322, P330, P361, P363, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
  |
Danger |
H300: Fatal if swallowed [Danger Acute toxicity, oral] H340: May cause genetic defects [Danger Germ cell mutagenicity] |
P201, P202, P264, P270, P281, P301+P310, P308+P313, P321, P330, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| (S)-N-(5,6,7,9-Tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo[a]heptalen-7-yl)acetamide | (S)-N-(5,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo[a]heptalen-7-yl) acetamide | (S)-colchicin |
| (S)-colchicina | (S)-colchicine | 078C484 |
| 24218-EP2275412A1 | 24218-EP2277865A1 | 24218-EP2281815A1 |
| 24218-EP2301933A1 | 24218-EP2301940A1 | 24218-EP2305219A1 |
| 24218-EP2305640A2 | 24218-EP2311827A1 | 24218-EP2314590A1 |
| 30512-31-3 | 4lzr | 4o2b |
| 64-86-8 | 7-alpha-H-Colchicine | 7.alpha.H-Colchicine |
| 7alphaH-Colchicine | A3324 | ACon1_000353 |
| AI3-31149 | AK167930 | AKOS001582887 |
| API0002098 | AS-13686 | Acetamide, N-((7S)-5,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo(a)heptalen-7-yl)- |
| Acetamide, N-(5,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo(a)heptalen-7-yl)-, (S)- | Acetamide, N-(5,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo(alpha)heptalen-7-yl)- | Acetamide, N-[(7S)-5,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo[a]heptalen-7-yl]- |
| Acetamide,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo(.alpha.)heptalen-7-yl)- | Acetamide,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo[.alpha.]heptalen-7-yl)- | BDBM50014846 |
| BG0155 | BPBio1_000535 | BRD-K00259736-001-06-5 |
| BRD-K00259736-001-10-7 | BSPBio_000485 | BSPBio_002083 |
| Benzo(a)heptalen-9(5H)-one | Benzo(a)heptalen-9(5H)-one, 7-acetamido-6,7-dihydro-1,2,3,10-tetramethoxy- | Benzo[a]heptalen-9(5H)-one,7-dihydro-1,2,3,10-tetramethoxy- |
| C 9754 | C-21700 | C-7100 |
| C07592 | CAS-64-86-8 | CC-25958 |
| CCG-39910 | CCRIS 691 | CHEBI:27882 |
| CHEMBL107 | CS-1141 | Certified Reference Material |
| Colchicin | Colchicin [German] | Colchicina |
| Colchicina [Italian] | Colchicine (JP17/USP) | Colchicine (TN) |
| Colchicine [JAN] | Colchicine [USP:JAN] | Colchicine for system suitability, European Pharmacopoeia (EP) Reference Standard |
| Colchicine, (European Pharmacopoeia (EP) Reference Standard) | Colchicine, >=95% (HPLC), powder | Colchicine, >=96.0% (HPLC) |
| Colchicine, BioReagent, plant cell culture tested, >=95% (HPLC) | Colchicine, Colchicum autumnale | Colchicine, Pharmaceutical Secondary Standard |
| Colchicine, United States Pharmacopeia (USP) Reference Standard | Colchicinum | Colchineos |
| Colchisol | Colchysat | Colcin |
| Colcrys | Colsaloid | Colstat |
| Condylon | D00570 | DB08117 |
| DSSTox_CID_4845 | DSSTox_GSID_24845 | DSSTox_RID_77551 |
| DTXSID5024845 | DivK1c_000753 | EBD2156734 |
| EC 200-598-5 | EINECS 200-598-5 | EU-0100310 |
| FT-0603187 | GTPL2367 | Goutnil |
| HMS1569I07 | HMS1920A08 | HMS2091G16 |
| HMS2096I07 | HMS2231C05 | HMS3260N22 |
| HMS3713I07 | HMS502F15 | HSDB 3044 |
| HY-16569 | IDI1_000753 | J10109 |
| KBio1_000753 | KBio2_001322 | KBio2_003890 |
| KBio2_006458 | KBio3_001303 | KBioGR_000856 |
| KBioSS_001322 | KM0881 | Kolkicin |
| LOC | LP00310 | LS-279 |
| Lopac0_000310 | M01514 | MCULE-1568647156 |
| MCULE-7858118731 | MEGxp0_001879 | MLS001055448 |
| MLS001055448-02 | MLS001304089 | MLS002153786 |
| MPC-004 | Mitigare | N-((7S)-5,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo(a)heptalen-7-yl)- acetamide |
| N-(5,6,7,9-Tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo(a)heptalen-7-yl)acetamide | N-(5,6,7,9-Tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo[.alpha.]heptalen-7-yl)-acetamide | N-Acetyl trimethylcolchicinic acid methylether |
| N-[(7S)-1,2,3,10-tetramethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl]acetamide | N-[(7S)-1,2,3,10-tetramethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl]acetamide; | N-[(7S)-1,2,3,10-tetramethoxy-9-oxo-6,7-dihydro-5H-benzo[a]heptalen-7-yl]acetamide |
| N-[(7S)-1,2,3,10-tetramethoxy-9-oxo-6,7-dihydro-5H-benzo[d]heptalen-7-yl]acetamide | N-[(7S)-5,6,7,9-tetrahydro-1,2,3,10-tetramethoxy-9-oxobenzo[a]heptalen-7-yl]acetamide | N-[(7s)-1,2,3,10-Tetramethoxy-9-Oxo-6,7-Dihydro-5h-Benzo[d]heptalen-7-Yl]ethanamide |
| N1721 | NCGC00025125-01 | NCGC00025125-02 |
| NCGC00025125-03 | NCGC00025125-04 | NCGC00025125-05 |
| NCGC00025125-06 | NCGC00025125-07 | NCGC00025125-08 |
| NCGC00025125-09 | NCGC00025125-10 | NCGC00025125-11 |
| NCGC00025125-12 | NCGC00025125-13 | NCGC00025125-14 |
| NCGC00025125-15 | NCGC00025125-18 | NCGC00025125-20 |
| NCGC00169157-01 | NCGC00169157-02 | NCGC00169157-03 |
| NCGC00254359-01 | NCGC00259096-01 | NCGC00260995-01 |
| NCI60_041659 | NINDS_000753 | NSC 757 |
| NSC-756702 | NSC-757 | NSC756702 |
| NSC757 | Pharmakon1600-01500205 | Prestwick0_000363 |
| Prestwick1_000363 | Prestwick2_000363 | Prestwick3_000363 |
| Prestwick_695 | SBI-0050298.P004 | SC-11404 |
| SCHEMBL8469 | SDCCGMLS-0066633.P001 | SDCCGSBI-0050298.P006 |
| SML2Y3J35T | SMR000058323 | SPBio_000289 |
| SPBio_002406 | SPECTRUM1500205 | SR-01000075794 |
| SR-01000075794-1 | SR-01000075794-3 | SR-01000075794-6 |
| SR-01000075794-7 | SR-01000597576 | SR-01000597576-1 |
| SR-01000597576-3 | Spectrum2_000075 | Spectrum3_000362 |
| Spectrum4_000298 | Spectrum5_000787 | Spectrum_000842 |
| Tocris-1364 | Tox21_110947 | Tox21_110947_1 |
| Tox21_201547 | Tox21_300582 | Tox21_500310 |
| UNII-SML2Y3J35T | UPCMLD-DP065 | UPCMLD-DP065:001 |
| WLN: L B677 MV&T&J CO1 DO1 EO1 JMV1 NO1 | ZINC621853 | ZX-AFC000369 |
| binds to tubulin | colchicine | inhibits microtubular assembly |
| methoxylated analogue of XD1 | s2284 | spindle poison |