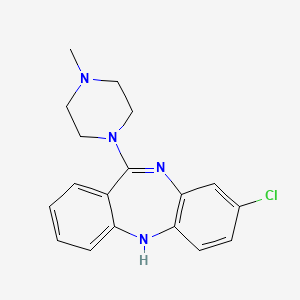
D0031 | Clozapine
N
N05AH02 Clozapine
[N05AH] Diazepines, oxazepines, thiazepines and oxepines
[N05A] ANTIPSYCHOTICS
[N05] PSYCHOLEPTICS
[N] Nervous system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| DEPOLARIZATION | Increase | 307 | ||||||
| OXYGEN CONSUMPTION RATE (OCR) | 300 μM | 2 minutes | human | HepG2 | Measurement of OCR | Negative | EC50 | 7 |
| ELECTRON TRANSPORT CHAIN | rat | isolated liver mitochondria | Negative | 23 | ||||
| ELECTRON TRANSPORT CHAIN | inhibit | 197 | ||||||
| ELECTRON TRANSPORT CHAIN | decrease | 307 | ||||||
| ECAR | 300 μM | 2 minutes | human | HepG2 | Measurement of ECAR | Negative | EC50 | 7 |
| SWELLING | Increase | 307 | ||||||
| SHAPE | 197 | |||||||
| Target | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| NADH:ubiquinone reductase | rat | isolated liver mitochondria | Negative | 23 | ||||
| Pyruvate kinase PKLR | Oxidation | 307 | ||||||
| Malate dehydrogenase, mitochondrial | Oxidation | 307 | ||||||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
  |
Danger |
Aggregated GHS information provided by 118 companies from 14 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H301 (100%): Toxic if swallowed [Danger Acute toxicity, oral] H341 (84.75%): Suspected of causing genetic defects [Warning Germ cell mutagenicity] H361 (88.14%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P264, P270, P281, P301+P310, P308+P313, P321, P330, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
  |
Danger |
H301: Toxic if swallowed [Danger Acute toxicity, oral] H341: Suspected of causing genetic defects [Warning Germ cell mutagenicity] H351: Suspected of causing cancer [Warning Carcinogenicity] H361: Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] |
P201, P202, P264, P270, P281, P301+P310, P308+P313, P321, P330, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 251mg/kg (251mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 7, Pg. 667, 1973. | |
| mouse | LD50 | subcutaneous | 194mg/kg (194mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 7, Pg. 667, 1973. | |
| women | TDLo | oral | 168mg/kg/4W-I (168mg/kg) | blood: agranulocytosis | Netherlands Journal of Medicine. Vol. 52, Pg. 26, 1998. |
| rat | LD50 | intramuscular | 210mg/kg (210mg/kg) | Farmaco, Edizione Pratica. Vol. 26, Pg. 585, 1971. | |
| guinea pig | LD50 | oral | 510mg/kg (510mg/kg) | Farmaco, Edizione Pratica. Vol. 26, Pg. 585, 1971. | |
| women | TDLo | oral | 280mg/kg/5W-I (280mg/kg) | American Journal of Psychiatry. Vol. 150, Pg. 985, 1993. | |
| man | TDLo | oral | 5357ug/kg/5D- (5.357mg/kg) | Journal of Clinical Pyschopharmacology. Vol. 12, Pg. 139, 1992. | |
| man | TDLo | oral | 3280mg/kg/82W (3280mg/kg) | blood: agranulocytosis | American Journal of Psychiatry. Vol. 153, Pg. 1503, 1996. |
| man | TDLo | oral | 86mg/kg/15D-I (86mg/kg) | British Journal of Psychiatry. Vol. 173, Pg. 440, 1998. | |
| rat | LD50 | subcutaneous | 240mg/kg (240mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 7, Pg. 667, 1973. | |
| man | TDLo | oral | 4286ug/kg/11D (4.286mg/kg) | Journal of Clinical Pyschopharmacology. Vol. 13, Pg. 155, 1993. | |
| women | TDLo | oral | 20mg/kg (20mg/kg) | Annals of Internal Medicine. Vol. 121, Pg. 722, 1994. | |
| man | TDLo | oral | 40mg/kg (40mg/kg) | Veterinary and Human Toxicology. Vol. 41, Pg. 20, 1999. | |
| mouse | LD50 | oral | 150mg/kg (150mg/kg) | Journal of Medicinal Chemistry. Vol. 23, Pg. 878, 1980. | |
| mouse | LD50 | intraperitoneal | 90mg/kg (90mg/kg) | Farmaco, Edizione Pratica. Vol. 26, Pg. 585, 1971. | |
| women | TDLo | oral | 28mg/kg/2W-I (28mg/kg) | gastrointestinal: changes in structure or function of endocrine pancreas | Lancet. Vol. 340, Pg. 251, 1992. |
| child | TDLo | oral | 4762ug/kg (4.762mg/kg) | American Journal of Emergency Medicine. Vol. 14, Pg. 462, 1996. | |
| man | TDLo | oral | 270mg/kg/6W-I (270mg/kg) | Annales de Medecine Interne. Vol. 144, Pg. 494, 1993. | |
| man | TDLo | oral | 27mg/kg/13D-I (27mg/kg) | Journal of Clinical Psychiatry. Vol. 55, Pg. 38, 1994. | |
| man | TDLo | oral | 2211mg/kg/37W (2211mg/kg) | blood: "changes in serum composition (e.g., tp, bilirubin, cholesterol)" | Journal of Clinical Pyschopharmacology. Vol. 15, Pg. 287, 1995. |
| rat | LD50 | intravenous | 41600ug/kg (41.6mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 7, Pg. 667, 1973. | |
| man | TDLo | oral | 429mg/kg (429mg/kg) | Journal of Toxicology, Clinical Toxicology. Vol. 38, Pg. 325, 2000. | |
| man | TDLo | oral | 429mg/kg/60D- (429mg/kg) | behavioral: muscle contraction or spasticity) | American Journal of Psychiatry. Vol. 152, Pg. 649, 1995. |
| mouse | LD50 | intravenous | 36500ug/kg (36.5mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 7, Pg. 667, 1973. | |
| human | TDLo | oral | 5mg/kg/7D-I (5mg/kg) | cardiac: pulse rate increase without fall in bp | Arzneimittel-Forschung. Drug Research. Vol. 22, Pg. 919, 1972. |
| women | TDLo | oral | 96mg/kg/48D-I (96mg/kg) | Human Psychopharmacology. Vol. 13, Pg. 583, 1998. | |
| women | TDLo | oral | 104mg/kg/26D- (104mg/kg) | blood: eosinophilia | Journal of Clinical Psychiatry. Vol. 59, Pg. 195, 1998. |
| dog | LD50 | oral | 145mg/kg (145mg/kg) | gastrointestinal: nausea or vomiting | Farmaco, Edizione Pratica. Vol. 26, Pg. 585, 1971. |
| Clozapine |