D0899 | docetaxel
L
L01CD02 Docetaxel
[L01CD] Taxanes
[L01C] PLANT ALKALOIDS AND OTHER NATURAL PRODUCTS
[L01] ANTINEOPLASTIC AGENTS
[L] Antineoplastic and immunomodulating agents
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| MEMBRANE POTENTIAL | 14.96±29.77 | human | qHTS-HepG2 | MMP assay | decrease | IC50 | 163 | |
| MEMBRANE POTENTIAL | human | HepG2 | MMP assay | Negative | IC50 | 163 | ||
| MEMBRANE POTENTIAL | 12.07±4.78 | rat | hepatocytes | MMP assay | decrease | IC50 | 163 | |
| FUSION | 185 | |||||||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
  |
Danger |
Aggregated GHS information provided by 10 companies from 8 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H319 (70%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H341 (80%): Suspected of causing genetic defects [Warning Germ cell mutagenicity] H351 (20%): Suspected of causing cancer [Warning Carcinogenicity] H360 (40%): May damage fertility or the unborn child [Danger Reproductive toxicity] H361 (50%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] H362 (20%): May cause harm to breast-fed children [Reproductive toxicity, effects on or via lactation] H372 (40%): Causes damage to organs through prolonged or repeated exposure [Danger Specific target organ toxicity, repeated exposure] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P260, P263, P264, P270, P280, P281, P305+P351+P338, P308+P313, P314, P337+P313, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
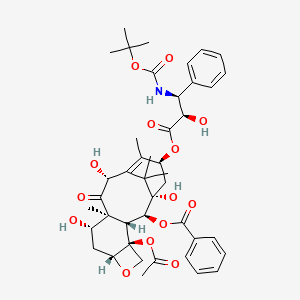
| (1R,2aR,4S,4aS,6R,9S,11S,12S,12aR)-1-Acetoxy-9-(((2R,3S)-3-((tert-butoxycarbonyl)amino)-2-hydroxy-3-phenylpropanoyl)oxy)-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-1H-7,11-methanocyclodeca[3,4]benzo[1,2-b]oxet-12-yl benzoate | (1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-(acetyloxy)-15-{[(2R,3S)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-3-phenylpropanoyl]oxy}-1,9,12-trihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0^{3,10}.0^{4,7}]heptadec-13-en-2-yl benzoate | (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl (aR,bS)-b-[[(1,1-dimethylethoxy)carbonyl]amino]-a-hydroxybenzenepropanoate |
| (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-12b-acetoxy-9-(((2R,3S)-3-((tert-butoxycarbonyl)amino)-2-hydroxy-3-phenylpropanoyl)oxy)-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-2a,3,4,4a,5,6,9,10 | (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-12b-acetoxy-9-(((2R,3S)-3-((tert-butoxycarbonyl)amino)-2-hydroxy-3-phenylpropanoyl)oxy)-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-1H-7,11-methanocyclodeca[3,4]benzo[1,2-b]oxet-12-yl benzoate | (2alpha,5beta,7beta,10beta,13alpha)-4-(acetyloxy)-13-({(2R,3S)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl}oxy)-1,7,10-trihydroxy-9-oxo-5,20-epoxytax-11-en-2-yl benzoate |
| (2beta,5beta,7alpha,8alpha,10alpha,13alpha)-4-(acetyloxy)-13-({(2R,3S)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl}oxy)-1,7,10-trihydroxy-9-oxo-5,20-epoxytax-11-en-2-yl benzoate | 114977-28-5 | 114977-28-5, Docetaxel |
| 4-(acetyloxy)-13alpha-({(2R,3S)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl}oxy)-1,7beta,10beta-trihydroxy-9-oxo-5beta,20-epoxytax-11-en-2alpha-yl benzoate | 5?,20-Epoxy-1,7?,10?-trihydroxy-9-oxotax-11-ene-2?,4,13?-triyl 4-acetate 2-benzoate 13-[(2R,3S)-3-[[(1,1-dimethylethoxy)carbonyl]amino]-2-hydroxy-3-phenylpropanoate] | 699121PHCA |
| AB0072965 | AB01273941-01 | AB01273941-02 |
| AC-383 | AK-72918 | AKOS015960718 |
| AKOS024457953 | ANX-514 | Anhydrous Docetaxel, European Pharmacopoeia (EP) Reference Standard |
| BDBM36351 | BRD-K30577245-001-04-3 | BRD-K30577245-341-01-9 |
| Benzenepropanoic acid, beta-(((1,1-dimethylethoxy)carbonyl)amino)-alpha-hydroxy-, (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca(3,4)benz(1,2-b)oxet-9-yl ester, (alphaR,betaS)- | Benzenepropanoic acid, beta-(((1,1-dimethylethoxy)carbonyl)amino)-alpha-hydroxy-, 12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca(3,4)benz(1,2-b)oxet-9-yl ester | Benzenepropanoic acid,1-dimethylethoxy)carbonyl]amino]-.alpha.-hydroxy-, (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-12b-(acetyloxy)-12-(benzyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester, (.alpha.R,.beta.S); |
| C11231 | CAS-114977-28-5 | CD-195 |
| CD0182 | CHEBI:4672 | CHEMBL92 |
| CID148124 | CS-1144 | D07866 |
| D4102 | DB01248 | DSSTox_CID_20464 |
| DSSTox_GSID_40464 | DSSTox_RID_79497 | DTXSID0040464 |
| Docecad | Docefrez | Docetaxel (JAN/INN) |
| Docetaxel (TN) | Docetaxel - Taxotere | Docetaxel Winthrop |
| Docetaxel [INN] | Docetaxel anhydrous | Docetaxel intermediate |
| Docetaxel(Taxotere) | Docetaxel, 98% | Docetaxel, Trihydrate |
| Docetaxel, purum, >=97.0% (HPLC) | Docetaxol | Docetaxolum |
| Docetaxolum [INN-Latin] | EX-A1206 | EmDOC |
| GTPL6809 | HMS2089K08 | HSDB 6965 |
| HY-B0011 | InChI=1/C43H53NO14/c1-22-26(55-37(51)32(48)30(24-15-11-9-12-16-24)44-38(52)58-39(3,4)5)20-43(53)35(56-36(50)25-17-13-10-14-18-25)33-41(8,34(49)31(47)29(22)40(43,6)7)27(46)19-28-42(33,21-54-28)57-23(2)45/h9-18,26-28,30-33,35,46-48,53H,19-21H2,1-8H3,(H,44,5 | KS-1452 |
| MCULE-1930158681 | MFCD00800737 | N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetylpaclitaxel |
| N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol | N-debenzoyl-N-Boc-10-deacetyl taxol | N-debenzoyl-N-tert-butoxycarbonyl-10-deacetyltaxol |
| NCGC00181306-01 | NCGC00181306-02 | NCGC00181306-04 |
| NCGC00242509-01 | NSC 628503 | NSC-628503 |
| NSC628503 | Q-100074 | Q420436 |
| RP 56976 | RP-56976 | RP56976 |
| SC-16998 | SCHEMBL4419 | SDP-014 |
| SID 530 | SR-01000003023 | SR-01000003023-5 |
| SYP-0704A | TXL | Taxoel |
| Taxotere | Taxotere (Aventis) | Taxotere (TN) |
| Taxotere(R) | Tox21_112781 | Tox21_112781_1 |
| Tox21_113088 | UNII-699121PHCA | W-1428 |
| W-60384 | XRP-6976L | Z1551429742 |
| ZDZOTLJHXYCWBA-VCVYQWHSSA- | ZDZOTLJHXYCWBA-VCVYQWHSSA-N | ZINC85537053 |
| [2aR-[2a?,4?,4a?,6?,9?(?R*,?S*),11?,12?,12a?,12b?]]-?-[[(1,1-Dimethylethoxy)carbonyl]amino]-?-hydroxy-12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester benzenepropanoic acid | [acetoxy-[(2R,3S)-3-(tert-butoxycarbonylamino)-2-hydroxy-3-phenyl-propanoyl]oxy-trihydroxy-tetramethyl-oxo-[?]yl] benzoate | bind-014 |
| docetaxel | docetaxel 114977-28-5 |