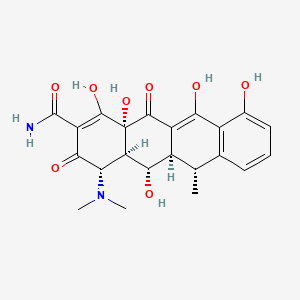
D1414 | doxycycline
J
A
J01AA20 Combinations of tetracyclines
[J01AA] Tetracyclines
[J01A] TETRACYCLINES
[J01] ANTIBACTERIALS FOR SYSTEMIC USE
[J] Antiinfectives for systemic use
J01AA02 Doxycycline
[J01AA] Tetracyclines
[J01A] TETRACYCLINES
[J01] ANTIBACTERIALS FOR SYSTEMIC USE
[J] Antiinfectives for systemic use
A01AB22 Doxycycline
[A01AB] Antiinfectives and antiseptics for local oral treatment
[A01A] STOMATOLOGICAL PREPARATIONS
[A01] STOMATOLOGICAL PREPARATIONS
[A] Alimentary tract and metabolism
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| MITOCHONDRIAL RESPIRATORY CHAIN COMPLEX ASSEMBLY | 15 µg/ml | 6 days | 143B osteosarcoma cells | BN PAGE analysis, In-gel complex I activity and western blotting | inhibitor | 285 | ||
| MITOCHONDRIAL PROTEIN TRANSLATION | 143B osteosarcoma cells | inhibitor | 285 | |||||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
  |
Danger |
Aggregated GHS information provided by 117 companies from 15 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H302 (74.36%): Harmful if swallowed [Warning Acute toxicity, oral] H315 (72.65%): Causes skin irritation [Warning Skin corrosion/irritation] H317 (10.26%): May cause an allergic skin reaction [Warning Sensitization, Skin] H319 (72.65%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H335 (72.65%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure Respiratory tract irritation] H360 (26.5%): May damage fertility or the unborn child [Danger Reproductive toxicity] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P261, P264, P270, P271, P272, P280, P281, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P321, P330, P332+P313, P333+P313, P337+P313, P362, P363, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
 |
Warning |
Aggregated GHS information provided by 96 companies from 4 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. Reported as not meeting GHS hazard criteria by 3 of 96 companies. For more detailed information, please visit ECHA C&L website Of the 3 notification(s) provided by 93 of 96 companies with hazard statement code(s): H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral] H312 (55.91%): Harmful in contact with skin [Warning Acute toxicity, dermal] H315 (59.14%): Causes skin irritation [Warning Skin corrosion/irritation] H319 (59.14%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H332 (55.91%): Harmful if inhaled [Warning Acute toxicity, inhalation] H335 (59.14%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure Respiratory tract irritation] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P305+P351+P338, P312, P321, P322, P330, P332+P313, P337+P313, P362, P363, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
 |
Warning |
Aggregated GHS information provided by 2 companies from 1 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral] H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation] H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] H335 (100%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure Respiratory tract irritation] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 241mg/kg (241mg/kg) | Pharmacological and Biochemical Properties of Drug Substances. Vol. 2, Pg. 305, 1979. | |
| mouse | LD50 | oral | 1870mg/kg (1870mg/kg) | Giornale Italiano di Chemioterapia. Italian Journal of Chemotherapy. Vol. 17, Pg. 276, 1970. | |
| dog | LD50 | intravenous | > 100mg/kg (100mg/kg) | Pharmacological and Biochemical Properties of Drug Substances. Vol. 2, Pg. 305, 1979. | |
| mouse | LD50 | intraperitoneal | 410mg/kg (410mg/kg) | Giornale Italiano di Chemioterapia. Italian Journal of Chemotherapy. Vol. 17, Pg. 276, 1970. | |
| rat | LD50 | oral | > 2gm/kg (2000mg/kg) | Pharmacological and Biochemical Properties of Drug Substances. Vol. 2, Pg. 305, 1979. | |
| rat | LD50 | intraperitoneal | 378mg/kg (378mg/kg) | Therapie. Vol. 23, Pg. 575, 1968. | |
| women | TDLo | oral | 68mg/kg/24D-I (68mg/kg) | musculoskeletal: joints | Postgraduate Medical Journal. Vol. 67, Pg. 313, 1991. |
| women | TDLo | oral | 68mg/kg/24D-I (68mg/kg) | Postgraduate Medical Journal. Vol. 67, Pg. 313, 1991. | |
| rat | LD50 | intravenous | 228mg/kg (228mg/kg) | Pharmacological and Biochemical Properties of Drug Substances. Vol. 2, Pg. 305, 1979. | |
| dog | LD50 | oral | > 500mg/kg (500mg/kg) | Pharmacological and Biochemical Properties of Drug Substances. Vol. 2, Pg. 305, 1979. | |