D0034 | Dantrolene
M
M03CA01 Dantrolene
[M03CA] Dantrolene and derivatives
[M03C] MUSCLE RELAXANTS, DIRECTLY ACTING AGENTS
[M03] MUSCLE RELAXANTS
[M] Musculoskeletal system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| ELECTRON TRANSPORT CHAIN | 50 μM | bovine | heart mitochondria | Measurement of complex I activity | Negative | p < 0.05 | 3 | |
| ELECTRON TRANSPORT CHAIN | 50 μM | bovine | heart mitochondria | Measurement of complex II + III activity | Negative | p < 0.05 | 3 | |
| ELECTRON TRANSPORT CHAIN | 50 μM | bovine | heart mitochondria | Measurement of complex II + III activity | Negative | p < 0.05 | 3 | |
| ELECTRON TRANSPORT CHAIN | 50 μM | bovine | heart mitochondria | Measurement of complex IV activity | Negative | p < 0.05 | 3 | |
| ELECTRON TRANSPORT CHAIN | 50 μM | bovine | heart mitochondria | Measurement of complex V activity | Negative | p < 0.05 | 3 | |
| Target | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| NADH:ubiquinone reductase | 50 μM | bovine | heart mitochondria | Measurement of complex I activity | Negative | p < 0.05 | 3 | |
| Succinate dehydrogenase | 50 μM | bovine | heart mitochondria | Measurement of complex II + III activity | Negative | p < 0.05 | 3 | |
| Quinol--cytochrome-c reductase | 50 μM | bovine | heart mitochondria | Measurement of complex II + III activity | Negative | p < 0.05 | 3 | |
| Cytochrome c oxidase | 50 μM | bovine | heart mitochondria | Measurement of complex IV activity | Negative | p < 0.05 | 3 | |
| ATP synthase | 50 μM | bovine | heart mitochondria | Measurement of complex V activity | Negative | p < 0.05 | 3 | |
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
 |
Warning |
Aggregated GHS information provided by 7 companies from 2 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. Reported as not meeting GHS hazard criteria by 1 of 7 companies. For more detailed information, please visit ECHA C&L website Of the 1 notification(s) provided by 6 of 7 companies with hazard statement code(s): H341 (100%): Suspected of causing genetic defects [Warning Germ cell mutagenicity] H351 (100%): Suspected of causing cancer [Warning Carcinogenicity] H361 (100%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] H373 (100%): Causes damage to organs through prolonged or repeated exposure [Warning Specific target organ toxicity, repeated exposure] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P260, P281, P308+P313, P314, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | subcutaneous | > 16gm/kg (16000mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 11, Pg. 3065, 1977. | |
| mouse | LD50 | intraperitoneal | 534mg/kg (534mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 11, Pg. 3065, 1977. | |
| rat | LD50 | intraperitoneal | 413mg/kg (413mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 11, Pg. 3065, 1977. | |
| mouse | LD50 | intravenous | > 50mg/kg (50mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 11, Pg. 3065, 1977. | |
| rat | LD50 | intravenous | > 50mg/kg (50mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 11, Pg. 3065, 1977. | |
| rat | LD50 | subcutaneous | > 16gm/kg (16000mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 11, Pg. 3065, 1977. | |
| women | LDLo | oral | 600mg/kg (600mg/kg) | New York State Journal of Medicine. Vol. 77, Pg. 1759, 1977. | |
| mouse | LD50 | intravenous | > 7gm/kg (7000mg/kg) | Journal of Medicinal Chemistry. Vol. 21, Pg. 127, 1978. | |
| human | TDLo | oral | 320mg/kg (320mg/kg) | JAMA, Journal of the American Medical Association. Vol. 231, Pg. 662, 1975. | |
| mouse | LD50 | oral | 1188mg/kg (1188mg/kg) | Journal of Pharmaceutical Sciences. Vol. 69, Pg. 327, 1980. | |
| rat | LD50 | oral | 7432mg/kg (7432mg/kg) | Kiso to Rinsho. Clinical Report. Vol. 11, Pg. 3065, 1977. | |
| dog | LD50 | oral | > 10550mg/kg (10550mg/kg) | Iyakuhin Kenkyu. Study of Medical Supplies. Vol. 25, Pg. 815, 1994. | |
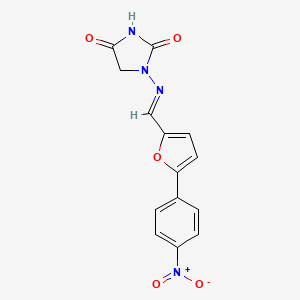
| (E)-1-(((5-(4-nitrophenyl)furan-2-yl)methylene)amino)imidazolidine-2,4-dione | 1-(((5-(4-Nitrophenyl)-2-furanyl)methylene)amino)-2,4-imidazolidinedione | 1-(((E)-[5-(4-Nitrophenyl)-2-furyl]methylidene)amino)-2,4-imidazolidinedione # |
| 1-((5-(p-Nitrophenyl)furfurylidene)amino)hydantoin | 1-({(1E)-[5-(4-nitrophenyl)furan-2-yl]methylidene}amino)imidazolidine-2,4-dione | 1-({[5-(4-nitrophenyl)furan-2-yl]methylidene}amino)imidazolidine-2,4-dione |
| 1-[5-(4-nitro-phenyl)-furan-2-ylmethyleneamino]-imidazolidine-2,4-dione | 1-[[5-(p-Nitrophenyl)furfurylidene]amino]hydantoin | 2,4-Imidazolidinedione, 1-(((5-(4-nitrophenyl)-2-furanyl)methylene)amino)- |
| 2,4-Imidazolidinedione, 1-[[[5-(4-nitrophenyl)-2-furanyl]methylene]amino]- | 7261-97-4 | 833480-90-3 |
| 85008-71-5 | A13928 | BCBcMAP01_000067 |
| BDBM50198767 | BIDD:GT0187 | BPBio1_000246 |
| BRD-K81272440-001-02-6 | BRD-K81272440-236-05-1 | BRN 0705189 |
| BSPBio_000222 | BSPBio_001305 | BSPBio_003074 |
| C06939 | C14H10N4O5 | CHEBI:4317 |
| CHEMBL1201288 | D02347 | DB01219 |
| DTXSID70873546 | Dantrium | Dantrolene (USAN/INN) |
| Dantrolene [USAN:BAN:INN] | Dantrolene [USAN:INN:BAN] | Dantroleno |
| Dantroleno [INN-Spanish] | Dantrolenum | Dantrolenum [INN-Latin] |
| EINECS 230-684-8 | F-368 | F64QU97QCR |
| HMS1361B07 | HMS1791B07 | HMS1989B07 |
| HMS3402B07 | HSDB 3050 | Hydantoin, 1-((5-(p-nitrophenyl)furfurylidene)amino)- |
| IDI1_000898 | IDI1_033775 | LS-76264 |
| Lopac0_000424 | NCGC00163132-01 | NCGC00163402-01 |
| NCGC00163402-02 | NCGC00163402-03 | NCGC00188950-01 |
| OZOMQRBLCMDCEG-VIZOYTHASA-N | Prestwick2_000291 | Prestwick3_000291 |
| R452 | Ryanodex | SCHEMBL12417164 |
| SCHEMBL40893 | SR-01000076241-8 | Spectrum5_001752 |
| UNII-F64QU97QCR | ZINC7997966 | [4-(5-{(E)-[(2,4-Dioxoimidazolidin-1-yl)imino]methyl}furan-2-yl)phenyl](hydroxy)oxoammonium |
| dantrolene |
| DrugBank Name | Dantrolene |
| DrugBank | DB01219 |
| CAS Number | 14663-23-1, 7261-97-4, 833480-90-3, 85008-71-5 |
| PubChem Compound | 6914273 |
| KEGG Compound ID | C06939 |
| KEGG Drug | D02347 |
| PubChem.Substance | 46504976 |
| ChEBI | 4317 |
| PharmGKB | PA449208 |
| ChemSpider | 5290202 |
| BindingDB | 50198767.0 |
| TTD | DNC001623 |
| Wikipedia | Dantrolene |
| DPD | 2402 |