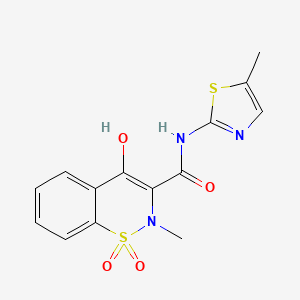
Drug
D0079 | Meloxicam
M
M01AC56 Meloxicam, combinations
[M01AC] Oxicams
[M01A] ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS, NON-STEROIDS
[M01] ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS
[M] Musculoskeletal system
M01AC06 Meloxicam
[M01AC] Oxicams
[M01A] ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS, NON-STEROIDS
[M01] ANTIINFLAMMATORY AND ANTIRHEUMATIC PRODUCTS
[M] Musculoskeletal system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| TRANSMEMBRANE POTENTIAL | 382 | 24hr | rat | hepatocytes | tetramethylrhodamine ethyl ester (TMRE) | decrease | AC50 (μM) | 40 |
| UNCOUPLING | 197 | |||||||
| MEMBRANE POTENTIAL | 14.47±0.00 | human | qHTS-HepG2 | MMP assay | decrease | IC50 | 163 | |
| MEMBRANE POTENTIAL | human | HepG2 | MMP assay | Negative | IC50 | 163 | ||
| MEMBRANE POTENTIAL | rat | hepatocytes | MMP assay | Negative | IC50 | 163 | ||
| STATE 2 RESPIRATION | 22.8 ± 2.8 | rat | isolated rat liver mitochondria | State 2 respiration ( 96-well plate format using a phosphorescent oxygen-sensitive probe MitoXpress) | inhibit | UC50 (nmol/mg mitochondrial protein) | 40 | |
| STATE 3 RESPIRATION | 100 nmol/mg mitochondrial protein | rat | isolated rat liver mitochondria | State 3 respiration ( 96-well plate format using a phosphorescent oxygen-sensitive probe MitoXpress) | Negative | IC50 (nmol/mg mitochondrial protein) | 40 | |
| LIPID METABOLISM | > 500 | 24hr | rat | hepatocytes | LipidTox, for neutral lipid accumulation, to evaluate lipid content. | accumulation | AC50 (μM) | 40 |
| GLUTATHIONE METABOLISM | 316 | 24hr | rat | hepatocytes | glutathion depletion: cells were incubated with 50 μM monochlorobimane with 6 μg/ml Hoechst 33342 | AC50 (μM) | 40 | |
| ROS PRODUCTION | NR | rat | hepatocytes | use CM-H2DCFDA to monitor reactive oxygen species | Negative | AC50 (μM) | 40 | |
| CYTOCHROME C RELEASE | 245 | 24hr | rat | hepatocytes | cytochrome c release (anti-cytochrome c antibody ) | induce | AC50 (μM) | 40 |
| ER STRESS-INDUCED | 153 | 24hr | rat | hepatocytes | DNA damage 153 induction (GADD153 antibodies) for ER-stress induced apoptosis | induce | AC50 (μM) | 40 |
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
 |
Danger |
H372: Causes damage to organs through prolonged or repeated exposure [Danger Specific target organ toxicity, repeated exposure] |
P260, P264, P270, P314, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| (3Z)-2-methyl-3-[[(5-methyl-1,3-thiazol-2-yl)amino]-oxidanyl-methylidene]-1,1-bis(oxidanylidene)-1$l^{6},2-benzothiazin-4-one | (3Z)-3-[hydroxy-[(5-methyl-2-thiazolyl)amino]methylidene]-2-methyl-1,1-dioxo-1$l^{6},2-benzothiazin-4-one | (E)-3-(hydroxy((5-methylthiazol-2-yl)amino)methylene)-2-methyl-2,3-dihydro-4H-benzo[e][1,2]thiazin-4-one 1,1-dioxide |
| 1027136-06-6 | 125M387 | 133687-22-6 |
| 2H-1,2-Benzothiazine-3-carboxamide, 4-hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-, 1,1-dioxide | 2H-1,2-Benzothiazine-3-carboxamide, 4-hydroxy-2-methyl-N-(5-methylthiazolyl)-, 1,1-dioxide | 3-{hydroxy[(5-methyl-1,3-thiazol-2-yl)amino]methylidene}-2-methyl-2,3-dihydro-4H-1,2-benzothiazin-4-one 1,1-dioxide |
| 4-Hydroxy-1,1-dioxo-1,2-dihydro-1lambda*6*-benzo[e][1,2]thiazine-3-carboxylic acid (5-methyl-thiazol-2-yl)-amide | 4-Hydroxy-2-methyl-1,1-dioxo-1,2-dihydro-1lambda*6*-benzo[e][1,2]thiazine-3-carboxylic acid (5-methyl-thiazol-2-yl)-amide | 4-Hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-2H-1,2-benzothiazin-3-crboxamide 1,1-dioxide |
| 4-Hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide | 4-Hydroxy-2-methyl-N-(5-methyl-2-thiazoyl)-2H-1,2-benzothiazine-3-carboxamide-1,1-dioxide | 4-Hydroxy-2-methyl-N-(5-methylthiazol-2-yl)-2H-benzo[e][1,2]thiazine-3-carboxamide 1,1-dioxide |
| 4-hydroxy-2-mehtyl-N-(5-mehtyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide | 4-hydroxy-2-methyl-N-(5-methy-2-thiazolyl)-2H-1,2-benzothiazine-3-caboxamide-1,1-dioxide | 4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-1,1-dioxo-1$l^{6},2-benzothiazine-3-carboxamide |
| 4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-1,1-dioxo-1?^{6},2-benzothiazine-3-carboxamide | 4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-1,1-dioxo-2H-1$l^{6},2-benzothiazine-3-carboxamide | 4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-1,1-dioxo-2H-1lambda6,2-benzothiazine-3-carboxamide |
| 4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide | 4-hydroxy-2-methyl-N-(5-methyl-2-thiazole)-2H-1,2-benzothiazine-3 -carboxamide 1,1-dioxide | 4-hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide-1,1-dioxide |
| 71125-38-7 | A837087 | AB0007297 |
| AB00383033-17 | AB00383033-18 | AB00383033_20 |
| AB1009494 | AC-1325 | AK-77454 |
| AKOS000279442 | AKOS026749959 | ALBB-027268 |
| AN-668/13244001 | ANW-57553 | BBL029076 |
| BB_SC-04648 | BCP11928 | BDBM50056998 |
| BG0248 | BIDD:GT0726 | BSPBio_002257 |
| C-20653 | C08169 | C14H13N3O4S2 |
| CAS-71125-38-7 | CC-30198 | CCG-39098 |
| CCRIS 9139 | CHEBI:6741 | CHEMBL1741042 |
| CHEMBL599 | CPD000718800 | CS-2247 |
| CTK6C3599 | CTK6C3600 | Coxflam |
| Coxicam | D00969 | DB-055490 |
| DB00814 | DSSTox_CID_803 | DSSTox_GSID_20803 |
| DSSTox_RID_75796 | DTXSID1020803 | EN300-52507 |
| ETI-511 | F2173-0387 | FT-0082769 |
| FT-0628193 | GTPL7220 | HMS1922D19 |
| HMS2089B18 | HMS2096I19 | HMS2234P07 |
| HMS2236C09 | HMS3259H06 | HMS3372C13 |
| HMS3372P18 | HMS3655K06 | HMS3713I19 |
| HMS3744K09 | HSCI1_000045 | HSDB 7741 |
| HY-B0261 | J10218 | KBio2_002113 |
| KBio2_004681 | KBio2_007249 | KBio3_001477 |
| KBioGR_001234 | KBioSS_002113 | KS-00000JN4 |
| KS-1084 | LS-40538 | Lopac0_000766 |
| M1959 | MCULE-4138373089 | MFCD00868752 |
| MLS000028587 | MLS001304725 | MLS001306413 |
| MLS006011422 | MXM | Melfax |
| Melonex | Meloxicam (JAN/USAN/INN) | Meloxicam (Mobic) |
| Meloxicam 1.0 mg/ml in Dimethyl Sulfoxide | Meloxicam [USAN:BAN:INN] | Meloxicam [USAN:USP:INN:BAN] |
| Meloxicam(Mobic) | Meloxicam, 99-101% | Meloxicam,(S) |
| Meloxicamum | Meloxicamum [Latin] | Meloxivet |
| Metacam | Mobec | Mobic |
| Mobic (TN) | Mobicox | Movalis |
| Movatec | N-1539 | NC00698 |
| NCGC00018248-01 | NCGC00018248-02 | NCGC00018248-03 |
| NCGC00018248-04 | NCGC00018248-05 | NCGC00018248-06 |
| NCGC00018248-07 | NCGC00018248-08 | NCGC00022924-03 |
| NCGC00022924-04 | NCGC00256316-01 | NCGC00259238-01 |
| NCGC00263878-02 | Opera_ID_2 | Parocin |
| Q414028 | R3928 | RTR-031220 |
| Revmoksikam | SAM002589991 | SC-12165 |
| SCHEMBL33369 | SCHEMBL3576 | SCHEMBL713100 |
| SMP2_000133 | SMR000058994 | SMR000718800 |
| SPBio_000902 | SPECTRUM1504150 | SR-01000003132-10 |
| ST24023559 | STK620505 | SW219562-1 |
| Spectrum2_000941 | Spectrum3_000649 | Spectrum4_000787 |
| Spectrum5_001813 | Spectrum_001633 | Tenaron |
| Tox21_111734 | Tox21_111734_1 | Tox21_113192 |
| Tox21_201689 | Tox21_302756 | UH-AC 62XX |
| UHAC-62XX | UNII-VG2QF83CGL | VG2QF83CGL |
| Vivlodex | Z1695493323 | ZINC103620661 |
| ZINC13129998 | ZRVUJXDFFKFLMG-UHFFFAOYSA-N | ZX-AN025777 |
| meloxicam | miloxicam | s1734 |
1. Dykens et al. (2007)
2. Moreno-Sanchez et al. (1999)
3. Vuda et al. (2016)