D0377 | Nitrofurantoin
J
J01XE51 Nitrofurantoin, combinations
[J01XE] Nitrofuran derivatives
[J01X] OTHER ANTIBACTERIALS
[J01] ANTIBACTERIALS FOR SYSTEMIC USE
[J] Antiinfectives for systemic use
J01XE01 Nitrofurantoin
[J01XE] Nitrofuran derivatives
[J01X] OTHER ANTIBACTERIALS
[J01] ANTIBACTERIALS FOR SYSTEMIC USE
[J] Antiinfectives for systemic use
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| UNCOUPLING | increase | 36 | ||||||
| MEMBRANE POTENTIAL | 26.31±6.35 | human | qHTS-HepG2 | MMP assay | decrease | IC50 | 163 | |
| MEMBRANE POTENTIAL | 8.69 | human | HepG2 | MMP assay | decrease | IC50 | 163 | |
| MEMBRANE POTENTIAL | 7.26±6.35 | rat | hepatocytes | MMP assay | decrease | IC50 | 163 | |
| MEMBRANE POTENTIAL | 442.5 µM | 30 mins | mouse | liver mitochondria | Rh123 fluorescence (excitation 485 nm, emission 535 nm) are recorded using a fluorescence multi-well plate reader (mCICCP (20 µM) treatments was considered as the 100% baseline for ΔΨm loss) | decrease | EC20 | 36 |
| RESPIRATION | 232.3 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Rotenone (2µM) was used as 100% baseline for complex I inhibition. | decrease | EC20 | 36 |
| RESPIRATION | 8.7 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Oligomycin A (1µM) was used as 100% baseline for complex II inhibition. | decrease | EC20 | 36 |
| STATE 3 RESPIRATION | 1 mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used glutamate as substrate | decrease | p < 0.05; 61% inhibition | 13 | |
| STATE 3 RESPIRATION | 50 μM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used glutamate as substrate | decrease | p < 0.05; 72% inhibition | 13 | |
| STATE 3 RESPIRATION | 5 μM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used glutamate as substrate | decrease | p < 0.05; 38% inhibition | 13 | |
| STATE 3 RESPIRATION | 1 mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used β-hydroxybutyrate as substrate | decrease | p < 0.05 | 13 | |
| STATE 3 RESPIRATION | 50 μM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used β-hydroxybutyrate as substrate | decrease | p < 0.05; 60% inhibition | 13 | |
| STATE 3 RESPIRATION | 5 μM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used β-hydroxybutyrate as substrate | decrease | p < 0.05 | 13 | |
| STATE 3 RESPIRATION | 1 mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used α-ketoghttarate as substrate | decrease | p < 0.05 | 13 | |
| STATE 3 RESPIRATION | 50 μM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used α-ketoghttarate as substrate | decrease | p < 0.05; 70% inhibition | 13 | |
| STATE 3 RESPIRATION | 5 μM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake; used α-ketoghttarate as substrate | decrease | p < 0.05 | 13 | |
| ELECTRON TRANSPORT CHAIN | decrease | 35 | ||||||
| DNP-UNCOUPLED RESPIRATION | 1 mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake | decrease | p < 0.05 | 13 | |
| DNP-UNCOUPLED RESPIRATION | 0.05mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake | Negative | p < 0.05 | 13 | |
| DNP-UNCOUPLED RESPIRATION | 0.005mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake | decrease | p < 0.05 | 13 | |
| DNP-UNCOUPLED RESPIRATION | 1 mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake | decrease | p < 0.05 | 13 | |
| DNP-UNCOUPLED RESPIRATION | 0.05mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake | decrease | p < 0.05 | 13 | |
| DNP-UNCOUPLED RESPIRATION | 1 mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake | decrease | p < 0.05 | 13 | |
| DNP-UNCOUPLED RESPIRATION | 0.05mM | mouse; C57B/6J | liver mitochondria | Measurement of oxygen uptake | decrease | p < 0.05 | 13 | |
| GLUCOSE GALACTOSE IC50 RATIO | permeabilized LUHMES cells | Assessment of the function of individual mitochondrial complexes using Agilent Seahorse XFe24 | Negative | EC25(NA) [Glc/Gal] | 326 | |||
| SWELLING | > 800 µM | 30 mins | mouse | liver mitochondria | swelling assay: Absorbance at 545 nm using a fluorescence multi-well plate reader (CaCl2 (50 µM) was considered as the 100% baseline for the swelling ) | increase | EC20 | 36 |
| Target | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| NADH:ubiquinone reductase | inhibitor | 35 | ||||||
| NADH:ubiquinone reductase | 232.3 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Rotenone (2µM) was used as 100% baseline for complex I inhibition. | inhibit | EC20 | 36 |
| Succinate dehydrogenase | 8.7 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Oligomycin A (1µM) was used as 100% baseline for complex II inhibition. | inhibit | EC20 | 36 |
| Cytochrome c | > 400 µM | 30 mins | mouse | liver mitochondria | Cytochrome c release was evaluated using ELISA kit ( 20 µg/ml Alamethicin was used as 100% baseline) | release | EC20 | 36 |
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
  |
Danger |
Aggregated GHS information provided by 91 companies from 15 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H302 (93.41%): Harmful if swallowed [Warning Acute toxicity, oral] H317 (58.24%): May cause an allergic skin reaction [Warning Sensitization, Skin] H334 (59.34%): May cause allergy or asthma symptoms or breathing difficulties if inhaled [Danger Sensitization, respiratory] H361 (10.99%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P261, P264, P270, P272, P280, P281, P285, P301+P312, P302+P352, P304+P341, P308+P313, P321, P330, P333+P313, P342+P311, P363, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 1493mg/kg (1493mg/kg) | Toxicology and Applied Pharmacology. Vol. 18, Pg. 185, 1971. | |
| rat | LD50 | subcutaneous | 178mg/kg (178mg/kg) | Toxicology and Applied Pharmacology. Vol. 18, Pg. 185, 1971. | |
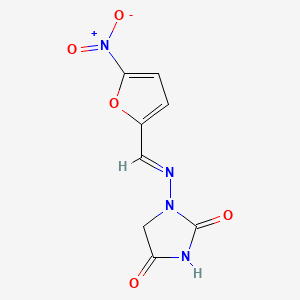
| (E)-1-(((5-Nitrofuran-2-yl)methylene)amino)imidazolidine-2,4-dione | (E)-1-[(5-nitro-2-furyl)methylideneamino]imidazolidine-2,4-dione | 1-(((5-Nitrofuran-2-yl)methylene)-amino)imidazolidine-2,4-dione |
| 1-(((5-Nitrofuran-2-yl)methylene)amino)imidazolidine-2,4-dione | 1-(((5-nitro-2-furanyl)methylene)amino)-2,4-imidazolidinedione | 1-((5-Nitrofurfurylidene)amino)hydantoin |
| 1-((5-nitro-2-furanyl)methylene)amino-2,4-imidazolidenedione | 1-((5-nitrofurfurylidene)amino)-hydantoin | 1-(5-Nitro-2-furfurylidenamino)hydantoin |
| 1-(5-Nitro-2-furfurylideneamino)hydantoin | 1-([(5-Nitro-2-furyl)methylidene]amino)-2,4-imidazolidinedione # | 1-[(1E)-2-(5-nitro(2-furyl))-1-azavinyl]-1,3-diazolidine-2,4-dione |
| 1-[(5-:nitrofurfurylidene)amino]hydantoin | 1-[(5-Nitrofurfurylidene)amino]hydantoin | 1-[(E)-(5-nitro-2-furanyl)methylideneamino]imidazolidine-2,4-dione |
| 1-[(E)-(5-nitro-2-furyl)methyleneamino]imidazolidine-2,4-dione | 1-[(E)-(5-nitrofuran-2-yl)methylideneamino]imidazolidine-2,4-dione | 1-[[(5-Nitro-2-furanyl)methylene]amino]-2,4-imidazolidinedione |
| 1-[[(5-nitrofuran-2-yl)methylene]amino]imidazolidine-2,4-dione | 1-{[(1E)-(5-nitro-2-furyl)methylene]amino}imidazolidine-2,4-dione | 1-{[(1E)-(5-nitrofuran-2-yl)methylidene]amino}imidazolidine-2,4-dione |
| 1-{[(5-nitro-2-furyl)methylene]amino}imidazolidine-2,4-dione | 1-{[(5-nitrofuran-2-yl)methylidene]amino}imidazolidine-2,4-dione | 1-{[(E)-(5-nitrofuran-2-yl)methylidene]amino}imidazolidine-2,4-dione |
| 1-{[(E)-1-(5-nitro-2-furyl)methylidene]amino}-1H-imidazole-2,4(3H,5H)-dione | 178170-37-1 | 2,4-Imidazolidenedione, 1-(((5-nitro-2-furanyl)methylene)amino)- |
| 2,4-Imidazolidinedione, 1-(((5-nitro-2-furanyl)methylene)amino)- | 2,4-Imidazolidinedione, 1-[[(5-nitro-2-furanyl)methylene]amino]- | 4-hydroxy-1-{[(E)-(5-nitrofuran-2-yl)methylidene]amino}-1,5-dihydro-2H-imidazol-2-one |
| 5-Nitrofurantoin | 5-Nitrofurantoindorn | 67-20-9 |
| 927AH8112L | A16008 | A835659 |
| AB00052052_03 | AB00513815 | AI3-26388 |
| AKOS001678301 | ARONIS27199 | Alfuran |
| BDBM57045 | BIDD:GT0181 | BPBio1_000039 |
| BRD-K76927775-001-05-0 | BSPBio_000035 | BSPBio_002073 |
| Benkfuran | Berkfuran | Berkfurin |
| C07268 | C8H6N4O5 | CCG-40108 |
| CCRIS 1192 | CHEBI:71415 | CHEBI:95222 |
| CHEMBL572 | CPD000058271 | Ceduran |
| Certified Reference Material | Chemiofuran | Cistofuran |
| Cyantin | Cystit | D00439 |
| DB00698 | Dantafur | EINECS 200-646-5 |
| Fua Med | Fuamed | Fur-ren |
| Furabid | Furachel | Furadantin |
| Furadantin (TN) | Furadantin Retard | Furadantina MC |
| Furadantine | Furadantine mc | Furadantine-MC |
| Furadantoin | Furadoin | Furadoine |
| Furadonin | Furadonine | Furadoninum |
| Furadontin | Furadoxyl | Furalan |
| Furaloid | Furantoin | Furantoina |
| Furatoin | Furedan | Furina |
| Furobactina | Furodantin | Furophen T |
| Furophen T-Caps | Gerofuran | HMS1568B17 |
| HMS1920P21 | HMS2091H16 | HMS2095B17 |
| HMS3712B17 | HMS500L06 | HSDB 3135 |
| HY-A0090 | Hydantoin, 1-((5-nitrofurfurylidene)amino)- | Hydantoin, 1-[(5-nitrofurfurylidene)amino]- |
| Hydantoin, 1-[(5-nitrofurfurylidene)amino]- (7CI,8CI) | Hydantoin, n-(5-nitro-2-furfurylidene)-1-amino- | IDI1_000224 |
| Ituran | Ivadantin | J01XE01 |
| J10193 | LS-1560 | MFCD00003224 |
| MLS000028500 | Macpac | Macrobid |
| Macrodantin | Macrodantina | Macrofuran |
| Macrofurin | N-(5-Nitro-2-furfurylidene)-1-aminohydantoin | N-(5-Nitro-2-furfurylideno)-1-aminohydantoina |
| N-(5-Nitro-2-furfurylideno)-1-aminohydantoina [Polish] | N-(5-Nitro-2-furfurylidine)-1-aminohydantoin | N-(5-Nitrofurfurylidene)-1-aminohydantoin |
| N-(5-Nitrofurfurylidene)-1aminohydantoin | N-(5-nitro-2-furfurylidene)-1-aminohyda ntoin | N-Toin |
| N0883 | NCGC00091505-01 | NCGC00091505-03 |
| NCGC00091505-04 | NCGC00091505-05 | NCGC00091505-06 |
| NCGC00091505-07 | NCGC00091505-08 | NCGC00091505-09 |
| NCGC00091505-10 | NCI-C55196 | ND-3320 |
| ND-7248 | NITROFURANTION | NITROFURANTOIN MACROCRYSTALLINE |
| NITROFURANTOIN, MACROCRYSTALLINE | NSC 2107 | NSC 44150 |
| NSC-2107 | NSC-757243 | NSC2107 |
| NSC44150 | NSC757243 | NXFQHRVNIOXGAQ-YCRREMRBSA-N |
| Nierofu | Nifurantin | Nifuretten |
| Nitoin | Nitrex | Nitrofur-C |
| Nitrofuradantin | Nitrofurantoin (JAN/USP/INN) | Nitrofurantoin Macro |
| Nitrofurantoin Macrocrystals | Nitrofurantoin [USAN:INN:BAN:JAN] | Nitrofurantoin [USP:INN:BAN:JAN] |
| Nitrofurantoin anhydrous | Nitrofurantoin macrocrystal | Nitrofurantoin, 97% |
| Nitrofurantoin, 98% | Nitrofurantoin, Antibiotic for Culture Media Use Only | Nitrofurantoin, Pharmaceutical Secondary Standard |
| Nitrofurantoin, United States Pharmacopeia (USP) Reference Standard | Nitrofurantoin, VETRANAL(TM), analytical standard | Nitrofurantoin, crystalline |
| Nitrofurantoin, macrocrystals | Nitrofurantoina | Nitrofurantoina [DCIT] |
| Nitrofurantoine | Nitrofurantoine [INN-French] | Nitrofurantoinum |
| Nitrofurantoinum [INN-Latin] | Nitrofurantoinum anhydrous | Novofuran |
| Orafuran | Parfuran | Pharmakon1600-01500433 |
| Phenurin | PiyEloseptyl | Prestwick2_000168 |
| Prestwick3_000168 | Prestwick_358 | PubChem6954 |
| Q-201479 | RTR-022602 | Ro-Antoin |
| SAM002554913 | SBB056985 | SBI-0051457.P003 |
| SC-06194 | SCHEMBL29470 | SCHEMBL29472 |
| SMR000058271 | SPECTRUM1500433 | SR-05000001681 |
| SR-05000001681-1 | SR-05000001681-2 | SR-05000001681-3 |
| SR-05000001681-4 | ST24031483 | STK009471 |
| STL454163 | Siraliden | Spectrum5_001367 |
| TR-022602 | Trantoin | UNII-927AH8112L |
| Uerineks | Urantoin | Urizept |
| Uro-Selz | Uro-Tablinen | Urodin |
| Urofuran | Urofurin | Urolisa |
| Urolong | Usaf ea-2 | Uvamin |
| Welfurin | Z-1941 | ZINC7997568 |
| Zoofurin | cid_4509 | fua-med |
| furophen | nitrofurantoin | s4536 |
| uro-tablineu | urodil |
| DrugBank Name | Nitrofurantoin |
| DrugBank | DB00698 |
| CAS Number | 178170-37-1, 54-87-5, 67-20-9 |
| PubChem Compound | 6604200 |
| KEGG Compound ID | C07268 |
| KEGG Drug | D00439 |
| PubChem.Substance | 46504447 |
| ChEBI | 71415 |
| PharmGKB | PA450640 |
| ChemSpider | 5036498 |
| BindingDB | 57045.0 |
| TTD | DAP000998 |
| Wikipedia | Nitrofurantoin |
| DPD | 10196 |