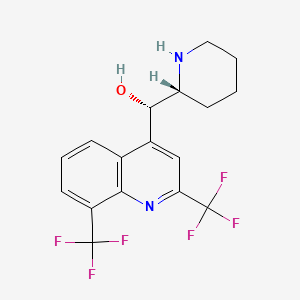
Drug
D0448 | mefloquine
P
P01BF02 Artesunate and mefloquine
[P01BF] Artemisinin and derivatives, combinations
[P01B] ANTIMALARIALS
[P01] ANTIPROTOZOALS
[P] Antiparasitic products, insecticides and repellents
P01BC02 Mefloquine
[P01BC] Methanolquinolines
[P01B] ANTIMALARIALS
[P01] ANTIPROTOZOALS
[P] Antiparasitic products, insecticides and repellents
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| ELECTRON TRANSPORT CHAIN | decrease | 35 | ||||||
| GLUCOSE GALACTOSE IC50 RATIO | 2.9 ± 1.1, 2.3 ± 1.0, 1.4 ,3.4 ± 1.3, 3.6 ± 2.2, 1.2 | 4hr | H9c2 cells | high-glucose–galactose cell viability assay with JC-1 mitochondrial membrane potential and ATP-depletion assays (CellTiter-Glo reagent ). | glucose/galactose IC50 ratio (JC-1 IC50 in glucose, JC-1 IC50 in galactose, JC-1 glu/gla, ATP IC50 in glucose, ATP IC50 in galactose, ATP glu/gla ) | 50 | ||
| Target | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| ATP synthase | inhibitor | 35 | ||||||
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
 |
Warning |
H302: Harmful if swallowed [Warning Acute toxicity, oral] |
P264, P270, P301+P312, P330, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| (+)-(11S,2'R)-erythro-Mefloquine | (+)-erythro-4-Quinolinemethanol, .alpha.-2-piperidinyl-2,8-bis(trifluoromethyl)- | (-)-(11S,2'R)-erythro-Mefloquine |
| (-)-Mefloquine | (DL-erythro-alpha-2-Piperidyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol | (S)-(2,8-bis(trifluoromethyl)quinolin-4-yl)((R)-piperidin-2-yl)methanol |
| (S)-[2,8-bis(trifluoromethyl)-4-quinolinyl]-[(2R)-2-piperidinyl]methanol;hydrochloride | (S)-[2,8-bis(trifluoromethyl)-4-quinolyl]-[(2R)-2-piperidyl]methanol | (S)-[2,8-bis(trifluoromethyl)-4-quinolyl]-[(2R)-2-piperidyl]methanol;hydrochloride |
| (S)-[2,8-bis(trifluoromethyl)quinolin-4-yl]-[(2R)-piperidin-2-yl]methanol;hydrochloride | (S)-[2,8-bis(trifluoromethyl)quinolin-4-yl][(2R)-piperidin-2-yl]methanol | (S,R)-MEFLOQUINE |
| 4-Quinolinemethanol, .alpha.-(2R)-2-piperidinyl-2,8-bis(trifluoromethyl)-, (.alpha.S)- | 4-Quinolinemethanol, alpha-(2R)-2-piperidinyl-2,8-bis(trifluoromethyl)-, (alphaS)-; | 4-Quinolinemethanol, alpha-(2R)-2-piperidinyl-2,8-bis(trifluoromethyl)-, (alphaS)-rel- |
| 4-Quinolinemethanol, alpha-2-piperidinyl-2,8-bis(trifluoromethyl)-, (R*,S*)-(+-)- | 51742-86-0 | 51742-87-1 |
| 53230-10-7 | BDBM50151865 | BDBM79170 |
| BIDD:GT0596 | BIDD:PXR0166 | BRD-K40645748-003-08-5 |
| C07633 | CHEBI:63687 | CHEMBL172 |
| D04895 | DTXSID4037168 | HSDB 6853 |
| LS-142023 | LS-187797 | MLS001332559 |
| MMV000014 | MMV000016 | Mefloquin |
| Mefloquina | Mefloquina [INN-Spanish] | Mefloquine (USAN/INN) |
| Mefloquine [USAN:INN:BAN] | Mefloquinum | Mefloquinum [INN-Latin] |
| Mephloquine | NCGC00161831-01 | NCGC00161831-02 |
| NCGC00161831-03 | Q27084116 | RO 13-7224 |
| RO 13-7225 | RTI1188-1-1 | RTI1189-1-1 |
| Racemic mefloquine | Ro 21-5998 | Ro 215998 |
| Ro-21-5998-001 | SCHEMBL21780 | SMR000875233 |
| UNII-TML814419R component XEEQGYMUWCZPDN-DOMZBBRYSA-N | WR 142,490 | WR 142490 |
| WR-142,490 | WR-177,602 | ZINC897085 |
| [(11S,2'R)-2,8-bis(trifluoromethyl)quinolin-4-yl]-(2-piperidyl)methanol | cid_65329 | mefloquine |