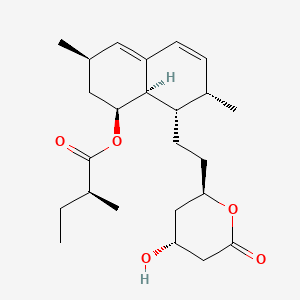
D0467 | lovastatin
C
C10BA01 Lovastatin and nicotinic acid
[C10BA] HMG CoA reductase inhibitors in combination with other lipid modifying agents
[C10B] LIPID MODIFYING AGENTS, COMBINATIONS
[C10] LIPID MODIFYING AGENTS
[C] Cardiovascular system
C10AA02 Lovastatin
[C10AA] HMG CoA reductase inhibitors
[C10A] LIPID MODIFYING AGENTS, PLAIN
[C10] LIPID MODIFYING AGENTS
[C] Cardiovascular system
| Toxicity | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| OPENING OF PERMEABILITY TRANSITION PORE (PTP) | 50 µM | 1 hour | Human | HepG2 | High-content screening assay | Decrease | MEC | 306 |
| UNCOUPLING | increase | 36 | ||||||
| MEMBRANE POTENTIAL | 43.6 µM | 30 mins | mouse | liver mitochondria | Rh123 fluorescence (excitation 485 nm, emission 535 nm) are recorded using a fluorescence multi-well plate reader (mCICCP (20 µM) treatments was considered as the 100% baseline for ΔΨm loss) | decrease | EC20 | 36 |
| MEMBRANE POTENTIAL | 100 µM | 1 hour | Human | HepG2 | High-content screening assay | Decrease | MEC | 306 |
| RESPIRATION | 4.4 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Rotenone (2µM) was used as 100% baseline for complex I inhibition. | decrease | EC20 | 36 |
| RESPIRATION | 6.2 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Oligomycin A (1µM) was used as 100% baseline for complex II inhibition. | decrease | EC20 | 36 |
| ELECTRON TRANSPORT CHAIN | decrease | 36 | ||||||
| ELECTRON TRANSPORT CHAIN | 100uM | C2C12 myoblasts | measured ubiquinol:cytochrome c oxidoreductase activity in broken C2C12 mitochondria after acute statin exposure at a fixed concentration for all compounds | decrease | 180 | |||
| S,N-GLYCEROPHOSPHATE SHUTTLE | C2C12 myoblasts | affect | 180 | |||||
| SWELLING | 71.5 µM | 30 mins | mouse | liver mitochondria | swelling assay: Absorbance at 545 nm using a fluorescence multi-well plate reader (CaCl2 (50 µM) was considered as the 100% baseline for the swelling ) | increase | EC20 | 36 |
| ROS PRODUCTION | 10 µM | 1 hour | Human | HepG2 | High-content screening assay | Increase | MEC | 306 |
| Target | Dose | Time | Species | Model | Method | Action | Positive criterion | Reference |
|---|---|---|---|---|---|---|---|---|
| NADH:ubiquinone reductase | inhibitor | 36 | ||||||
| NADH:ubiquinone reductase | 4.4 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Rotenone (2µM) was used as 100% baseline for complex I inhibition. | inhibit | EC20 | 36 |
| Succinate dehydrogenase | 6.2 µM | 60 mins | mouse | liver mitochondria | Oxygen consumption was monitored with 50nM MitoXpress ( an oxygen-sensitive phosphorescent dye) using a spectrofluorimeter (Tecan Infinite 200; λExcitation 380nm; λEmission 650nm). Oligomycin A (1µM) was used as 100% baseline for complex II inhibition. | inhibit | EC20 | 36 |
| Quinol--cytochrome-c reductase | 100uM | C2C12 myoblasts | measured ubiquinol:cytochrome c oxidoreductase activity in broken C2C12 mitochondria after acute statin exposure at a fixed concentration for all compounds | inhibitor | 180 | |||
| Glycerol-3-phosphate dehydrogenase, mitochondrial | C2C12 myoblasts | inhibitor | 180 | |||||
| Reactive oxygen species | 10 µM | 1 hour | Human | HepG2 | High-content screening assay | increase | MEC | 306 |
| Cytochrome c | 118.2 µM | 30 mins | mouse | liver mitochondria | Cytochrome c release was evaluated using ELISA kit ( 20 µg/ml Alamethicin was used as 100% baseline) | release | EC20 | 36 |
| Pictogram | Signal | Statements | Precautionary Statement Codes |
|---|---|---|---|
  |
Danger |
Aggregated GHS information provided by 142 companies from 13 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. Reported as not meeting GHS hazard criteria by 1 of 142 companies. For more detailed information, please visit ECHA C&L website Of the 12 notification(s) provided by 141 of 142 companies with hazard statement code(s): H302 (31.21%): Harmful if swallowed [Warning Acute toxicity, oral] H361 (67.38%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity] H372 (24.82%): Causes damage to organs through prolonged or repeated exposure [Danger Specific target organ toxicity, repeated exposure] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
P201, P202, P260, P264, P270, P281, P301+P312, P308+P313, P314, P330, P405, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
 |
Warning |
The GHS information provided by 1 company from 1 notification to the ECHA C&L Inventory. H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral] H312 (100%): Harmful in contact with skin [Warning Acute toxicity, dermal] H332 (100%): Harmful if inhaled [Warning Acute toxicity, inhalation] |
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P312, P322, P330, P363, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
 |
Warning |
H302: Harmful if swallowed [Warning Acute toxicity, oral] |
P264, P270, P301+P312, P330, and P501; (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Organism | Test type | Route | Dose (normalized dose) | Effect | Source |
|---|---|---|---|---|---|
| child | TDLo | oral | 17mg/kg/3W-I (17mg/kg) | behavioral: wakefulness | Lancet. Vol. 343, Pg. 973, 1994. |
| man | TDLo | oral | 240mg/kg/60W- (240mg/kg) | behavioral: muscle weakness | Annals of Pharmacotherpy. Vol. 26, Pg. 190, 1992. |
| human | TDLo | oral | 8750ug/kg/14D (8.75mg/kg) | Clinical Pharmacology and Therapeutics Vol. 50, Pg. 730, 1991. | |
| women | TDLo | oral | 285mg/kg/30W- (285mg/kg) | Israel Journal of Medical Sciences. Vol. 28, Pg. 101, 1992. | |
| mouse | LD50 | oral | > 1gm/kg (1000mg/kg) | Journal of Antibiotics. Vol. 32, Pg. 852, 1979. | |
| (+)-mevinolin | (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-Hexahydro-3,7-dimethyl-8-(2-(2R,4R)-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl (S)-2-methyl-butyrate | (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-Hexahydro-8-(2-((4R,6R)-4-hydroxy-2-oxo-2H-pyran-6-yl)ethyl)-3,7-dimethylnaphtyl(S)-2-methylbutyrat |
| (1S,3R,7S,8S,8aR)-8-(2-((2R,4R)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl)ethyl)-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (S)-2-methylbutanoate | (1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (2S)-2-methylbutanoate | (1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (2S)-2-methylbutanoate |
| (1S-(1alpha(R*),3alpha,7beta,8beta(2S*,4S*),8abeta))-2-Methylbutanoic acid 1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester | (2S)-(1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-Hexahydro-3,7-dimethyl-8-[2-[(2R,4R)-tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl]ethyl]-1-naphthalenyl-2-methyl butanoate | (2S)-2-Methylbutanoic acid (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-[(2R,4R)-tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl]ethyl]-1-naphthalenyl ester |
| (2S)-2-methylbutanoic acid [(1S,3R,7S,8S,8aR)-8-[2-[(2R,4R)-4-hydroxy-6-oxo-2-oxanyl]ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] ester | (S)-((1S,3R,7S,8S,8AR)-8-(2-((2R,4R)-4-HYDROXY-6-OXO-TETRAHYDRO-2H-PYRAN-2-YL)ETHYL)-3,7-DIMETHYL-1,2,3,7,8,8A-HEXAHYDRONAPHTHALEN-1-YL) 2-METHYLBUTANOATE | (S)-((1S,3R,7S,8S,8aR)-8-(2-((2R,4R)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl)ethyl)-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl) 2-methylbutanoate |
| (S)-2-Methyl-butyric acid (1S,3R,7S,8S,8aR)-8-[2-((3R,5R)-4-hydroxy-6-oxo-tetrahydro-pyran-2-yl)-ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl ester | (S)-2-Methylbutyric acid, 8-ester with (4R,6R)-6-(2-((1S,2S,6R,8S,8aR)-1,2,6,7,8,8a-hexahydro-8-hydroxy-2,6-dimethyl-1-naphthyl)ethyl)tetrahydro-4-hydroxy-2H-pyran-2-one | 1,2,6,7,8,8a-Hexahydro-beta,delta-dihydroxy-2,6-dimethyl-8-(2-methyl-1-oxobutyoxy)-1-naphthaleneheptanoic acid delta-lactone |
| 117141-EP2272825A2 | 117141-EP2298776A1 | 1S,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (2S)-2-methylbutanoate |
| 1cqp | 2-Methyl-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl]-1-naphthalenyl ester butanoic acid | 28048-EP2269989A1 |
| 28048-EP2270011A1 | 28048-EP2272825A2 | 28048-EP2272841A1 |
| 28048-EP2277507A1 | 28048-EP2277865A1 | 28048-EP2280006A1 |
| 28048-EP2281813A1 | 28048-EP2284158A1 | 28048-EP2286795A1 |
| 28048-EP2287165A2 | 28048-EP2287166A2 | 28048-EP2289892A1 |
| 28048-EP2292620A2 | 28048-EP2295406A1 | 28048-EP2295409A1 |
| 28048-EP2298742A1 | 28048-EP2298745A1 | 28048-EP2298772A1 |
| 28048-EP2298776A1 | 28048-EP2298779A1 | 28048-EP2301923A1 |
| 28048-EP2301931A1 | 28048-EP2301936A1 | 28048-EP2305648A1 |
| 28048-EP2308839A1 | 28048-EP2311808A1 | 28048-EP2311829A1 |
| 28048-EP2314588A1 | 2beta,6alpha-Dimethyl-8alpha-(2-methyl-1-oxobutoxy)-mevinic acid lactone | 330L755 |
| 6 alpha-Methylcompactin | 6-Methylcompactin | 6-alpha-Methylcompactin |
| 6alpha-Methylcompactin | 74133-25-8 | 75330-75-5 |
| 8-[2-((2R,4R)-4-hydroxy-6-oxo(2H-3,4,5-trihydropyran-2-yl))ethyl](1S,7S,8S,3R, 8aR)-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthyl (2S)-2-methylbutanoate | 96638-EP2287163A1 | 96638-EP2305678A1 |
| 9LHU78OQFD | A838030 | AB00052400-17 |
| AB00052400_18 | AB00052400_19 | AB0108514 |
| AC-13961 | ACT02620 | ACon0_000534 |
| ACon1_000390 | AKOS005267139 | ALBB-027272 |
| ANW-41686 | AOB5269 | ARONIS24208 |
| Altocor | Altoprev | Artein |
| BBL024473 | BB_NC-01457 | BDBM34168 |
| BG0243 | BIDD:GT0749 | BIDD:PXR0113 |
| BPBio1_000519 | BRD-K09416995-001-06-8 | BRD-K09416995-001-21-7 |
| BRN 3631989 | BSPBio_000471 | BSPBio_001265 |
| BSPBio_003346 | Belvas | Butanoic acid, 2-methyl-, (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-((2R,4R)-tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester, (2S)- |
| Butanoic acid, 2-methyl-, (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester, (2S)- | Butanoic acid, 2-methyl-, 1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-(tetrahydro-4-hydroxy-6-- oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester, (1S-(1alpha(R*),3alpha,7beta,8beta(2S*,4S*),8abeta))- | Butanoic acid, 2-methyl-, 1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester, (1S-(1-alpha-(R*),3-alpha,7-beta,8-beta-(2S*,4S*),8a-beta))- |
| Butanoic acid, 2-methyl-, 1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester, (1S-(1alpha(R*),3alpha,7beta,8beta(2S*,4S*),8abeta))- | Butanoic acid, 2-methyl-,1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester,(1S-(1alpha(R*),3alpha,7beta,8beta(2S*,4S*),8abeta)); | C-21799 |
| C07074 | C10AA02 | C24H36O5 |
| CAS-75330-75-5 | CC-30040 | CC-30041 |
| CCG-39627 | CCRIS 8092 | CHEBI:40303 |
| CHEMBL503 | CPD000673570 | CS-1990 |
| CTK2H7081 | Cholestra | Closterol |
| Colevix | D00359 | DB00227 |
| DSSTox_CID_784 | DSSTox_GSID_20784 | DSSTox_RID_75788 |
| DTXSID5020784 | DivK1c_001032 | EBD2126857 |
| EC 616-212-7 | EN300-52515 | G226 |
| GTPL2739 | HMS1569H13 | HMS1792O07 |
| HMS1923O13 | HMS1990O07 | HMS2089M06 |
| HMS2093O03 | HMS2096H13 | HMS2236F07 |
| HMS3039N16 | HMS3259F10 | HMS3268C03 |
| HMS3403O07 | HMS3412H19 | HMS3676H19 |
| HMS3713H13 | HMS503O05 | HSDB 6534 |
| HY-N0504 | Hipolip | Hipovastin |
| IDI1_001032 | J10136 | KBio1_001032 |
| KBio3_002848 | KS-1082 | KSC377A8D |
| L-154803 | L0214 | LOVALIP |
| LS-46359 | Lestatin | Lipdip |
| Lipivas | Lipofren | Liposcler |
| Lovalip | Lovalord | Lovastatin & Primycin |
| Lovastatin (MK-803) | Lovastatin (Mevacor) | Lovastatin (USP/INN) |
| Lovastatin [USAN:BAN:INN] | Lovastatin [USAN:USP:INN:BAN] | Lovastatin [USAN] |
| Lovastatin, 98% | Lovastatin,(S) | Lovastatina |
| Lovastatina [Spanish] | Lovastatine | Lovastatine [French] |
| Lovastatinum | Lovastatinum [Latin] | Lovasterol |
| Lovastin | Lozutin | MCULE-4740518260 |
| MCULE-7087866108 | MEGxm0_000398 | MEVACOR |
| MFCD00072164 | MK 803 | MK-803 |
| MK-803 | ML-530B | MLS000069585 |
| MLS001055358 | MLS006011867 | MSD 803 |
| Mevacor | Mevacor (TN) | Mevinacor |
| Mevinolin (lovastatin) | Mevinolin from Aspergillus sp., >=98% (HPLC) | Mevinolin from Aspergillus sp., powder |
| Mevlor | Monacolin K | Monakolin K |
| N1632 | NC00713 | NCGC00023509-03 |
| NCGC00023509-04 | NCGC00023509-05 | NCGC00023509-06 |
| NCGC00023509-07 | NCGC00023509-08 | NCGC00023509-09 |
| NCGC00023509-10 | NCGC00023509-11 | NCGC00023509-13 |
| NCGC00023509-14 | NCGC00023509-16 | NCGC00254157-01 |
| NCGC00259026-01 | NINDS_001032 | NSC-633781 |
| NSC-758662 | NSC633781 | NSC758662 |
| Nergadan | Opera_ID_1578 | PCZOHLXUXFIOCF-BXMDZJJMSA-N |
| Paschol | Pharmakon1600-01503977 | Prestwick0_000516 |
| Prestwick1_000516 | Prestwick2_000516 | Prestwick3_000516 |
| Prestwick_819 | Q417740 | RTR-031803 |
| Rextat | Rodatin | Rovacor |
| S-7779 | SAM002589904 | SAM002589963 |
| SBB080686 | SBI-0051881.P002 | SC-11968 |
| SCHEMBL14227102 | SCHEMBL3136 | SMR000058779 |
| SMR000673570 | SPBio_002392 | SPECTRUM1503977 |
| SR-01000000123 | SR-01000000123-3 | SR-05000001880 |
| SR-05000001880-1 | SR-05000001880-2 | STK801953 |
| Simvastatin impurity, lovastatin- | Sivlor | Spectrum3_001873 |
| Spectrum5_001294 | Taucor | Tecnolip |
| Teroltrat | Tox21_110888 | Tox21_110888_1 |
| Tox21_201475 | Tox21_300268 | UNII-9LHU78OQFD |
| US9115116, lovastatin | US9353061, Lovastatina | Z1258578375 |
| ZINC3812841 | [(1S,3R,7S,8S,8aR)-3,7-dimethyl-8-[2-[(2R,4R)-4-oxidanyl-6-oxidanylidene-oxan-2-yl]ethyl]-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] (2S)-2-methylbutanoate | [(1S,3R,7S,8S,8aR)-8-[2-[(2R,4R)-4-hydroxy-6-oxo-tetrahydropyran-2-yl]ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] (2S)-2-methylbutanoate |
| [(1S,3R,7S,8S,8aR)-8-[2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] (2S)-2-methylbutanoate | butanoic acid, 2-methyl-, (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-[(2R,4R)-tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl]ethyl]-1-naphthalenyl ester, (2S)- | cid_53232 |
| lovastatin | lovastatin-mevacor | mevinolin |
| s2061 |